Shi-Min Yuan
DOI: 10.5935/1678-9741.20140087
BNP: Brain natriuretic peptide
CK-MB: Creatine kinase isoenzyme MB
NT-proBNP: N-terminal pro-B-type natriuretic peptide
NYHA: New York Heart Association
POD: Postoperative day
POH: Postoperative hour
INTRODUCTION
Troponins are a type of contractile protein produced in cardiac and skeletal muscles. They are composed of troponin subunits I, C and T, with the former two subunits being highly sensitive and specific for myocardial damage, especially implicated in the diagnosis of acute coronary syndrome[1]. Dynamics of troponins have been sufficiently described as to start to increase a few hours after myocardial ischemic damage, reach a plateau after 10-15 hours, and recover gradually to the baseline level[2]. Elevations of troponins T and I were also found in patients without myocardial injury. These situations included severe renal dysfunction[3], acute neurological events (with a peak troponin I value of 3.43±3.22 µg/L)[4], hepatic cirrhosis and portal hypertension[5], stroke, pulmonary embolism and sepsis[6].
Troponins were sporadically reported in cardiac myxoma patients. However, these results were actually anecdotal without being taken into a through investigation. It was supposed that troponins in cardiac myxoma patients might be in a same fashion as in the conditions without myocardial injury. In order to verify this hypothesis, a patient with cardiac myxoma, whose clinical and histopathological aspects have been reported elsewhere[7,8], is to be presented in terms of her serum troponin studies along with creatine kinase isoenzyme MB (CK-MB) and N-terminal pro-B-type natriuretic peptide (NT-proBNP) by incorporating the results based on a comprehensive retrieval of the literature.
CLINICAL OBSERVATIONS
Six months prior to current admission, a 51-year-old female had abrupt loss of vision and consciousness. She was in a state of cardiogenic shock, which was revealed to be a result of multiple cerebral infarcts with a diagnosis of "top of the basilar" syndrome due to a left atrial myxoma. Later, she also developed multiple segmental embolism of her lower extremities involving the bilateral anterior tibial, bilateral posterior tibial, right popliteal and bilateral dorsal arteries. Left atrial myxoma resection was performed under cardiopulmonary bypass. During the operation, the myxoma was found to be originated from the free wall of the left atrium. The myxoma was resected en bloc, and the iatrogenic atrial free wall defect was repaired with an autologous pericardial patch. She had a delayed awakening until postoperative hour (POH) 13.5 without any new neurological sequelea. She had an uneventful postoperative course and was discharged home on postoperative day (POD) 19. Histopathology of the resected myxoma showed a glandular cardiac myxoma.
On admission, her C-reaction protein was 41.66 (normal range, 0-10) mg/L, hypersensitive C-reaction protein was >5 (normal range, 0-1) mg/L, and CK-MB 7.6 (normal range, 0-40) U/L. Her condition was gradually stable after prolonged treatment. She was recently referred to this hospital for surgical treatment of left atrial myxoma. After admission, the diagnosis of left atrial myxoma was confirmed by echocardiography and chest computed tomography. Her preoperative troponin I and NT-proBNP values were normal.
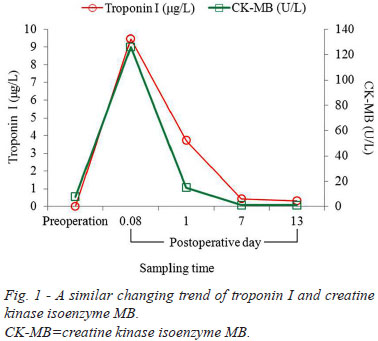
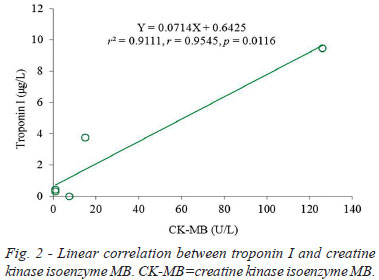
Postoperative detections revealed elevated troponin I and CK-MB and normal NT-proBNP (Table 1). Troponin I values were moderately elevated at POH 2 and POD 1. Postoperative CK-MB showed a same trend with cardiac troponin I, which reached a peak value at POH 2 (POD 0.08), gradually decreased on POH 24 (POD 1), and reached a plateau on PODs 7 and 13. The only difference between the two indicators was CK-MB recovered to normal values since POD 7, whereas troponin I did not, but were 0.43 µg/L and 0.32 µg/L at PODs 7 and 13 (Figure 1). A significant correlation could be noted between the postoperative values of the two indicators (Y=0.0714X + 0.6425, r2=0.9111, r=0.9545, P=0.0116) (Figure 2). Random NT-proBNP values were 243 and 448 pg/mL at POH 2 and on POD 7. No significant linear correlation between troponin I and NT-proBNP were found (Y=-0.0251X + 11.2760, r2=0.2846, r=-0.5335, P=0.6418) (Figure 3).

DISCUSSION
Troponin I is a popularly recognized sensitive biomarker in the diagnosis of postoperative myocardial damage and perioperative myocardial infarction[9]. Clinical observations revealed that troponin I, CK and CK-MB values peaked between POHs 6 and 18, troponin T between POHs 18 and 42, and myoglobin at the surgical closure. The values of all markers were higher in patients undergoing coronary surgery compared to those undergoing valve surgery[10]. Yuan et al.[11] discovered significant elevated serum troponin I during the early days after the onset or surgical repair of myocardial rupture, and they advocated the troponin I was valuable in judging the prognosis of the patients with myocardial injury. Troponin I (as well as NT-BNP) can be helpful in the evaluation of the functions of the vital organs including heart subjected to perioperative drug use, such as oral prophylactic metoprolol[12] and high-dose aprotinin[13].
Troponin levels in cardiac myxoma were reported in 18 sporadic patients[14-31], totally 19 patients including the present one (Table 2). Of them, 12 (63.2%) were troponin I and 7 (36.8%) were troponin T. Six (31.6%) patients were uncomplicated, and 13 (68.4%) were complicated (χ2=5.16, P=0.0502, Fisher's exact test). Of the complicated patients, acute myocardial infarction developed in 10 (76.9%) patients, and ventricular fibrillation arrest, pulmonary embolism, and ischemic stroke developed in 1 (7.7%) patient, each (χ2=21.17, P=0.0001, Fisher's exact test). The uncomplicated patients had a normal or only slightly elevated troponin before open heart surgery. However, the complicated patients showed a normal preoperative troponin in 3 (23.1%) and an elevated troponin in 10 (76.9%) patients (χ2=7.54, P=0.0169, Fisher's exact test). The overall quantitative result of preoperative troponin I was 2.45±2.53 (range, 0.0189-5.91; median, 1.47) µg/L (n=7), and that of troponin T was 3.10±4.29 (range, 0.31-9.37; median, 1.36) mg/L (n=4), respectively. The troponin I was somewhat higher than the baseline value of coronary artery disease patients (1.0±6 µg/L)[9] or the baseline value of the patients with coronary and/or valvular disorders (0.16±0.05 µg/L)[10] reported in the literature. The functional atrioventricular valve obstruction and embolic phenomena predisposing to myocardial infarction in the presence of a cardiac myxoma might be the explanations of the remarkable rise of the baseline troponin values[25].
It was proposed that troponin I levels measured on POD 1 may predict short-, medium- and long-term mortality. Troponin I levels of alive patients was 2.01 (range, 1.01-3.65) and 4.26 (range, 2.18-8.37) µg/L at POH 2 and on POD 1, while those of the deceased were 3.63 (range, 1.62-8.26) and 11.85 (range, 5.59-23.96) µg/L, respectively[14]. In a patient with a cardiac myxoma and associated coronary artery stenosis, his troponin I was 3.56 µg/L on POD 13[26]. The present patient with old cerebral infarct had an uneventful recovery after the resection of cardiac myxoma. She had compatible troponin I values during the early postoperative period to the maximal value of the survived surgical patients as reported by Croal et al.[32]. In addition, this patient had significantly decreased troponin I values on PODs 7 and 13, much lower than those of the patient reported by Stępień's et al.[26]. This could be interpreted by the associated coronary artery disease in the latter patient[26].
Comparative studies between troponins and CK-MB have already been stated some decades ago. There were little differences among troponins I and T, and CK-MB after cardiac operation to diagnose myocardial damage[33]. However, troponins have a more prolonged release than CK-MB when the patient is subjected to myocardial damage; it is therefore more sensitive than CK-MB in indicating myocardial necrosis. A troponin I concentration <15 µg/L within PODs 1-2 was highly suggestive of the absence of perioperative myocardial necrosis[34]. The dynamic changes of troponin I and CK-MB in the present patient were quite identical. The incidental finding was a peak value at POH 2 and a gradual decrease on POD 1, contrary to a delayed peak appearing on POD 1 as reported by Croal et al.[32]. The disconformity warranted further investigations.
NT-proBNP is a pre-propetptide synthetized in the cardiomyocytes. When it is secreted, it splits into two parts: physiologically active brain natriuretic peptide (BNP) and biologically inactive NT-proBNP. Both can be significantly elevated when the patient is with an acute or congestive heart failure[35]. The normal range of BNP was <200 pg/mL[36]. Increased BNP values were discovered to be related to acute cerebrovascular events[37]. BNP >140 pg/mL could be helpful in distinguishing cardiogenic from non-cardiogenic embolism[38]. Clinical observations revealed BNP levels were loosely correlated with New York Heart Association (NYHA) functional class[36]. In the patients undergoing cardiac surgery including one patient with atrial myxoma, BNP was 62.8±68.1 pg/mL[39]. Patients with congestive heart failure had a mean BNP concentration of 1076±138 pg/mL, while the non-congestive heart failure patients had a mean BNP concentration of 38±4 pg/mL[40]. Links between BNP and cardiac myxoma have rarely been discussed. Factors associated with high BNP levels other than congestive heart failure were age, gender, renal failure, lung disease and myocardial infarction; meanwhile, atrial myxoma was determined as a factor accounting for low BNP levels with congestive heart failure[41]. Plasma BNP level in 17 consecutive adult patients (including one patient with a left atrial myxoma) during the perioperative period were detected. Plasma BNP remained unchanged until 12, 24 and 48 hours after the termination of cardiopulmonary bypass, which were 149.5±43.0 pg/mL, 175.2±93.6 pg/mL and 146.2±59.4 pg/mL, respectively[42]. In a patient with right heart enlargement and severe pulmonary hypertension free of congestive heart failure, her BNP was 5,613.4 pg/mL[30]. A 53-year-old man with a large left atrial myxoma presented with biventricular heart failure. His BNP was as high as 4,800 pg/mL[43]. Lack of elevations of BNP has been reported in the event of acute congestive heart failure or with ventricular inflow obstruction (hypertrophic obstructive cardiomyopathy, mitral stenosis, or atrial myxoma)[36].
Top of the basilar artery refers to a 2-cm territory surrounding the five-forked junction at the top of the basilar artery formed by 2 posterior cerebral arteries, 2 superior cerebellar arteries and the top of basilar artery[44]. Cerebral infarction involving the rostral brainstem and cerebral hemisphere that are supplied by the distal basilar artery causes a clinical syndrome, known as rostral brainstem infarction or "top of the basilar" syndrome, characterized by visual, oculomotor, and behavioral abnormalities, often with insignificant motor dysfunction[45]. "Top of the basilar" syndrome occurs when the top of the basilar artery is subjected to thromboembolic occlusions, and results in bilateral thalamic ischemia due to occlusion of perforator vessels. Transient ischemic attack and stroke can present in 80% of patients with a cardiac myxoma[46]. Unexplained transient ischemic attack, cerebral infarction and syncope are common neurologic manifestations[47]. Clinical observations demonstrated that the patients with preoperative cerebral infarct took longer time to awaken than those without (6.5±5.0 hours vs. 4.6±2.9 hours, P<0.05)[48]. The incidence of confusion and cerebral infarction after operation in the preoperative infarct patients was higher than those without[48]. The anesthetic awareness in the present patient was 13.5 hours, even longer than reported. This phenomenon implicated that an old cerebral infarct may interfere anesthetic awareness, but not necessarily complicated with new postoperative sequelae. The normal postoperative BNP values of the present patient offered a strong support to this argument.
CONCLUSION
Troponins are not necessarily elevated in patients with a cardiac myxoma without coronary syndrome. By contrast, cardiac myxoma patients with an elevated troponin may herald the presence of an associated coronary event. An old cerebral infarct does not necessarily cause an elevation of troponin or BNP, or new neurological events, but might lead to a delayed awakening.
REFERENCES
1. Lum G, Solarz DE, Farney L. False positive cardiac troponin results in patients without acute myocardial infarction. Lab Med. 2006;37(9):546-50.
2. Hammarsten O. Small changes in cardiac troponin levels are common in patients with myocardial infarction: diagnostic implications. Conf Pap Med. 2013;2013:Article ID 583175, 5 pages.
3. Hamm CW, Giannitsis E, Katus HA. Cardiac troponin elevations in patients without acute coronary syndrome. Circulation. 2002;106(23):2871-2. [MedLine]
4. Dixit S, Castle M, Velu RP, Swisher L, Hodge C, Jaffe AS. Cardiac involvement in patients with acute neurologic disease: confirmation with cardiac troponin I. Arch Intern Med. 2000;160(20):3153-8. [MedLine]
5. Wiese S, Mortensen C, Gøtze JP, Christensen E, Andersen O, Bendtsen F, et al. Cardiac and proinflammatory markers predict prognosis in cirrhosis. Liver Int. 2014;34(6):e19-30. [MedLine]
6. Agewall S, Giannitsis E, Jernberg T, Katus H. Troponin elevation in coronary vs. non-coronary disease. Eur Heart J. 2011;32(4):404-11. [MedLine]
7. Yuan SM. Cerebral infarction due to cardiogenic emboli originating from atrial myxoma: a case report. Changhua J Med. 2014;12(2):87-92.
8. Yuan SM. Glandular cardiac myxoma: case report with literature review. Folia Morphol (Warsz). 2014;73(3):374-82. [MedLine]
9. Oliveira MA, Botelho PH, Brandi AC, Santos CA, Soares MJ, Zaiantchick M, et al. The importance of troponin I in the diagnosis of myocardial infarction in the postoperative of coronary artery bypass graft surgery. Rev Bras Cir Cardiovasc. 2009;24(1):11-4. [MedLine] View article
10. Di Stefano S, Casquero E, Bustamante R, Gualis J, Carrascal Y, Bustamante J, et al. Plasma troponins as markers of myocardial damage during cardiac surgery with extracorporeal circulation. Tohoku J Exp Med. 2007;213(1):63-9. [MedLine]
11. Yuan SM, Jing H, Lavee J. The implications of serum enzymes and coagulation activities in postinfarction myocardial rupture. Rev Bras Cir Cardiovasc. 2011;26(1):7-14. [MedLine] View article
12. Rossi Neto JM, Gun C, Ramos RF, Almeida AF, Issa M, Amato VL, et al. Myocardial protection with prophylactic oral metoprolol during coronary artery bypass grafting surgery: evaluation by troponin I. Rev Bras Cir Cardiovasc. 2013;28(4):449-54. [MedLine] View article
13. Ferreira CA, Vicente WV, Evora PR, Rodrigues AJ, Klamt JG, Carlotti AP, et al. High-dose aprotinin does not affect troponin I, N-Terminal pro-B-type natriuretic peptid and renal function in children submitted to surgical correction with extracorporeal circulation. Rev Bras Cir Cardiovasc. 2009;24(4):519-32. [MedLine] View article
14. Gurlertop Y, Yilmaz M, Erdogan F, Acikel M, Kose N. Left ventricular outflow tract myxoma. Eur J Echocardiogr. 2003;4(4):339-41. [MedLine]
15. Demir M, Akpinar O, Acarturk E. Atrial myxoma: an unusual cause of myocardial infarction. Tex Heart Inst J. 2005;32(3):445-7. [MedLine]
16. Balami JS, Jones HW, Alp N, Dwight J, Casser C, Martin A, et al. Atrial myxoma presenting as transient ischaemic attack and acute coronary syndrome in an octogenarian. Age Ageing. 2006;35(6):644. [MedLine]
17. Özdoğru İ, Duran M, Şarlı B, Oğuzhan A. Left atrial myxoma supplied by the circumflex coronary artery arising from the right sinus of Valsalva. Türk Kardiyol Dern Arş. 2008;36(8):549-51. [MedLine]
18. Yadav S, Alvarez JM. Catastrophic presentation of atrial myxoma with total occlusion of abdominal aorta. Interact Cardiovasc Thorac Surg. 2009;9(5):913-5. [MedLine]
19. Dalzell JR, Jackson CE, Castagno D, Gardner RS. Histologically benign but clinically malignant: an unusual case of recurrent atrial myxoma. QJM. 2009;102(3):229-30. [MedLine]
20. Albouaini K, Boghossian S, Alkarmi A. Myocardial infarction as an alert to the presence of left atrial myxoma. Eur J Gen Med. 2010;7(4):440-2.
21. Shimada Y, Maeda K, Ogawa K, Oe H, Matsumura Y, Yoshiyama M. Three dimensional echocardiography of tako-tsubo cardiomyopathy with atrial myxoma. BMJ Case Rep. 2010;2010. pii: bcr0720080544. [MedLine]
22. Agarwal S. A 22-year-old woman with ischemic stroke and left ventricular dysfunction. Case Presentation - October 2011 [Cited Nov 6 2014]. Available from: http://my.clevelandclinic.org/heart/medical-professionals/case-studies/ischemic-stroke.aspx
23. Vogel B, Thomas D, Mereles D, Rottbauer W, Katus HA. Systemic embolization and myocardial infarction due to clinically unrecognized left atrial myxoma. Case Rep Med. 2011;2011:159024. [MedLine]
24. Weiss SL, Russell HM, Lay A, Backer CL. Calcified right atrial myxoma in an adolescent. World J Pediatr Congenit Heart Surg. 2011;2(3):523-5. [MedLine]
25. Marta L, Peres M, Alves M, Ferreira da Silva G. Giant left atrial myxoma presenting as acute myocardial infarction. Rev Port Cardiol. 2012;31(12):815-9. [MedLine]
26. Stępień E, Grudzień G, Andres M, Jakóbczyk M, Czapczak D, Kapusta P, et al. A new clonal chromosomal aberration (47, XY, +21) in atrial myxoma from an elderly male patient. Cardiogenetics. 2012;2:e3.
27. Haffner PAM, Neves VCM, Lins G, Torres EML, Martins WA, et al. Myocardial infarction and myxoma in the left atrium. Arq Bras Cardiol: imagem cardiovasc. 2013;26(4):315-9.
28. Jung J, Hong YS, Lee CJ, Lim SH, Choi H, Lee S. Successful surgical treatment of a right atrial myxoma complicated by pulmonary embolism. Korean J Thorac Cardiovasc Surg. 2013;46(1):63-7. [MedLine]
29. Lazaros G, Masoura C, Brili S, Stavropoulos G, Kafiri G, Stefanadis C. Large left atrial myxoma in an oligosymptomatic young woman. Hellenic J Cardiol. 2013;54(1):60-3. [MedLine]
30. Leo S, Yang K, Weng C, Liang Z. Large atrial myxoma mimicking severe mitral stenosis associated with right heart enlargement and severe pulmonary hypertension. Cardiovasc Diagn Ther. 2013;3(1):52-4. [MedLine]
31. Sadeghpour A, Alizadehasl A. Showering emboli of an atrial mass: a fatal phenomenon. Res Cardiovasc Med. 2013;2(2):77-8. [MedLine]
32. Croal BL, Hillis GS, Gibson PH, Fazal MT, El-Shafei H, Gibson G, et al. Relationship between postoperative cardiac troponin I levels and outcome of cardiac surgery. Circulation. 2006;114(14):1468-75. [MedLine]
33. Bonnefoy E, Filley S, Kirkorian G, Guidollet J, Roriz R, Robin J, et al. Troponin I, troponin T, or creatine kinase-MB to detect perioperative myocardial damage after coronary artery bypass surgery. Chest. 1998;114(2):482-6. [MedLine]
34. Alyanakian MA, Dehoux M, Chatel D, Seguret C, Desmonts JM, Durand G, et al. Cardiac troponin I in diagnosis of perioperative myocardial infarction after cardiac surgery. J Cardiothorac Vasc Anesth. 1998;12(3):288-94. [MedLine]
35. Palazzuoli A, Gallotta M, Quatrini I, Nuti R. Natriuretic peptides (BNP and NT-proBNP): measurement and relevance in heart failure. Vasc Health Risk Manag. 2010;6:411-8. [MedLine]
36. B-type natriuretic peptide (BNP), plasma [Cited Nov 6 2014]. Available from: http://www.mayomedicallaboratories.com/test-catalog/Clinical+and+Interpretive/83873
37. Kim SH, Lee JY, Park SH, Jang HC, Lim EJ, Chang SJ, et al. Plasma B-type natriuretic peptide level in patients with acute cerebral infarction according to infarction subtype and infarction volume. Int J Med Sci. 2013;10(1):103-9. [MedLine]
38. Sakai K, Shibazaki K, Kimura K, Aoki J, Kobayashi K, Fujii S, et al. Brain natriuretic peptide as a predictor of cardioembolism in acute ischemic stroke patients: brain natriuretic peptide stroke prospective study. Eur Neurol. 2013;69(4):246-51. [MedLine]
39. Doyama K, Fukumoto M, Takemura G, Tanaka M, Oda T, Hasegawa K, et al. Expression and distribution of brain natriuretic peptide in human right atria. J Am Coll Cardiol. 1998;32(7):1832-8. [MedLine]
40. Maisel A. B-type natriuretic peptide measurements in diagnosing congestive heart failure in the dyspneic emergency department patient. Rev Cardiovasc Med. 2002;3 Suppl 4:S10-7. [MedLine]
41. Ahn MS, Yoo BS. Serial monitoring of B-type natriuretic peptide in heart failure patients. Korean Circ J. 2007;37(9):393-8.
42. Morimoto K, Ishiguro S, Kuroda H. Release of brain natriuretic peptide during the perioperative period of cardiac surgery. Yonago Acta Med. 1997;40(1):1-11.
43. Hodkinson EC, Dixon LJ, Dalzell GW. Multimodal imaging of an atypical left atrial myxoma. QJM. 2015;108(1):51-3. [MedLine]
44. Sato M, Tanaka S, Kohama A. "Top of the basilar" syndrome: clinico-radiological evaluation. Neuroradiology. 1987;29(4):354-9. [MedLine]
45. Caplan LR. "Top of the basilar" syndrome. Neurology. 1980;30(1):72-9. [MedLine]
46. Ugurlu B, Oto Ö, Okutan H, Kutluk K, Silistreli E, Sariosmanoğlu N, et al. Stroke and myxoma. Asian Cardiovasc Thorac Ann. 2000;8(2):130-3.
47. Yufe R, Karpati G, Carpenter S. Cardiac myxoma: a diagnostic challenge for the neurologist. Neurology. 1976;26(11):1060-5. [MedLine]
48. Cao L, Bi Q, Zhang Z. Neurologic dysfunction in the patients complicated with cerebral infarction after cardiac surgery. Beijing Med. 2002;24(5):301-3.
No financial support.
Author role & responsibility
SMY: Main Author
Article receive on Friday, March 28, 2014
 All scientific articles published at rbccv.org.br are licensed under a Creative Commons license
All scientific articles published at rbccv.org.br are licensed under a Creative Commons license