Chun-Li SongI; Qian LiI; Yun-Peng YuII; Guan WangII; Jin-Peng WangI; Yang LuII; Ji-Chang ZhangII; Hong-Ying DiaoI; Jian-Gen LiuI; Yi-Hang LiuI; Jia LiuI; Ying LiI; Dan CaiI; Bin LiuII
DOI: 10.5935/1678-9741.20150016
BSA: Bovine serum albumin
CAD: Coronary artery disease
DESs: Drug eluting stents
EDC: N-(3-dimethylaminopropyl)-N'-ethylcarbodiimide
ISR: In-stent restenosis
NHS: N-hydroxysuccinimide
VEGF: Vascular endothelial growth factor
INTRODUCTION
In-stent restenosis (ISR) was mainly caused by complications of intracoronary stent placement, including thromboembolic events and neointimal hyperplasia due to smooth muscle cell hyperproliferation. Drug eluting stents (DESs) have been designed mainly to reduce cellular proliferation and thus reduce ISR. Drug-eluting stents currently on the market release cytotoxic drugs such as paclitaxel and rapamycin to inhibit neointimal hyperplasia at the expense of delaying endothelialization[1,2].However, the incomplete endothelialization of the stent surface has been suggested that may lead to the increased long-term incidence of thrombosis and ISR[3].The critical role of the vascular endothelium in preventing thrombosis and regulating neointimal hyperplasia has resulted in restenosis prevention strategies that focus on enhancing endothelialiazation[4-6].
Vascular epithelial growth factor (VEGF), a cytokine originally described in 1983[7], is involved in processes essential to the growth, maintenance and repair of vascular structures. Exogenous VEGF has been reported to show accelerated re-endothelialization of damaged arteries in the rat carotid artery and attenuated intimal hyperplasia[8]. The delivery of VEGF using VEGF-eluting stents showed that it has been used to promote revascularization and re-endothelialization by stimulating endothelial progenitor cell migration and maturation [9,10]. Circulating endothelial progenitor cells (EPCs), a subset of bone marrow-derived stem cells, possess the ability to differentiate into functional and mature endothelial cells and recently have been identified as a key factor for re-endothelialization[11]. The EPC capture stents have been developed using immobilized antibodies targeted at EPC surface antigens, such as CD34[12].
In this report, to further accelerate re-endothelialization, we aimed to develop method to simultaneously coat VEGF and anti-CD34 antibody. Our results showed that VEGF and anti-CD34 antibody were successfully coated onto the 316 stainless steel.
Experimental procedures
Preparation of the coated steel sheet
Round 316L stainless steel sheets (diameter 6 mm, thickness 1 mm) were used to facilitate the measurement and evaluation of the properties of the coating, instead of bare metal stents with limited testable aspects and relatively high costs. The metal surface was polished, washed and dried at 60ºC for 24 hours, then sterilized by ultraviolet radiation. Heparin was conjugated to dopamine using N-(3-dimethylaminopropyl)-N'-ethylcarbodiimide (EDC) and N-hydroxysuccinimide (NHS)[13].The sterilized sheet was polymerized with compounds generated from condensation reaction of dopamine (Aladdin, Shanghai, China) and heparin (Aladdin) as described[13], and termed as the D-H group.
Then, sixteen sheets from the D-H group were immersed into 1ug/ml VEGF165 (Life technologies) dissolved in phosphate-buffered saline (PBS, PH 7.4) for 30 min at room temperature (RT). Washed with Milli-Q water for 3 times (5 min per time) and dried under nitrogen. Then, a Milli-Q water solution of heparin sodium (3mg/ml) was subsequently dip-coated for 30 min at RT. Washed with Milli-Q water for 3 times (5 min per time) and dried under nitrogen. These coating procedure were repeated 10 times, and these sixteen steel disks were set as the D-(H-V)10 group.
The D-(H-V)10-A group was obtained by the following procedures. Eight steel disks from the D-(H-V)10 group were immersed into 0.1mg/ml protein A (Aladdin) dissolved in PBS for 30 min at RT, blocked in 10 mg/ml bovine serum albumin (BSA; BD Biosciences) for 24 hours at 4 ºC and immersed with 2 µg/ml rabbit anti-human CD34 antibody (Abcam, Cambridge, UK) for 24 hours at 4 ºC. After the whole procedure, three groups of sheets were obtained (8 sheets/each group) for further evaluations.
Immunofluorescence detection of the coated VEGF and anti-CD34 antibody
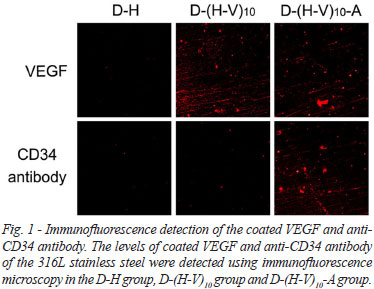
The levels of coated VEGF and anti-CD34 antibody of the stainless steel sheets were detected using immunofluorescence microscopy in the D-H group, D-(H-V)10 group and D-(H-V)10-A group. The sheets were soaked for 1 hour in blocking solution (PBS containing 3% BSA). For the detection of VEGF, all the three groups were incubated with rabbit anti-human VEGF primary antibodies (1:100; Abcam) overnight at 4 ºC, washed three times with PBS, and then incubated with Alexa Fluor® 488 Goat Anti-Rabbit IgG (Life technologies) at room temperature for 1 hour. For the detection of rabbit anti-human CD34 antibody, secondary antibodies- Alexa Fluor® 488 Goat Anti-Rabbit IgG were directly incubated. Wash three times to remove non-specific binding of the secondary antibodies and observe using a Laser scanning confocal microscope (Leica TCS SP5; Leica Microsystems, Germany).
The detection of coated VEGF and anti-CD34 antibody by ELISA
Coated steel sheets from the D-H group, D-(H-V)10 group and D-(H-V)10-A group were immersed into RIPA lysis buffer for 24 hours at 4 ºC to dissolve VEGF and anti-CD34 antibody. The levels of VEGF and anti-CD34 antibody were detected using VEGF165 ELISA Kit (Life technologies) and Rabbit IgG ELISA Kit (Novus Biologicals, USA), respectively.
Statistical analysis
Statistical significance was evaluated by comparing mean values (±standard deviation) using the two-tailed Student's t-test for independent groups. The probability value P<0.05 was considered to be statistically significant.
RESULTS
Immunofluorescence staining
To evaluate whether the 316L stainless steel sheets were successfully coated with VEGF and anti-CD34 antibody, immunofluorescence assay was performed. The results of immunofluorescence assay showed that VEGF could be detected in the D-(H-V)10 and D-(H-V)10-A group, suggesting the steel sheets were successfully covered with VEGF. Anti-CD34 antibody could only be observed in the D-(H-V)10-A group, which was the only group that coated with CD34 antibody (Figure 1). Our findings suggested that the 316L stainless steel sheets were successfully coated with VEGF and anti-CD34 antibody.
ELISA detection of coated VEGF and anti-CD34 antibody
To further evaluate the coverage of VEGF and anti-CD34 antibody, ELISA was performed in the D-H group, D-(H-V)10 group and D-(H-V)10-A group. Similar to the results of immunofluorescence assay, VEGF could be detected in the D-(H-V)10 and D-(H-V)10-A group, and anti-CD34 antibody could only be observed in the D-(H-V)10-A group (Figure 2). The results of ELISA further confirmed that we successfully coated VEGF and anti-CD34 antibody onto the 316 stainless steel.

DISCUSSION
Intravascular coronary stenting has been widely used for many years, and it has increased the quality of life and life expectancy of patients with coronary disease. The 316 stainless steel is one of the most widely used materials for coronary stents with a board range of mechanical properties. However, the exposure of flowing blood to the bare metal stent may lead to thrombus formation and smooth muscle cell proliferation, and finally cause in-stent restenosis (ISR).Therefore, huge amount of recent work has attempted to develop non-thrombogenic coating for these metallic stents[14-16].
Early drug-eluting stents coating with various kind of drugs have been designed to reduce the restenosis through minimize vascular inflammation and cellular proliferation[17], which including a polymer-based drug delivery platform and a pharmacologic agent (usually an immunosuppressant and/or antiproliferative compound).Though early trials seem to be exciting with markedly reduction rates of ISR (5%-8%)[18,19], long-term follow-up studies showed that DESs implantation increased the long-term risk of thrombosis by 15%-35% compared with bare-metal stents implantation[20].
Based on our expanding understanding of pathophysiology of restenosis, novel stent coating strategies have been developed, such as delivery of VEGF (e.g., VEGF-eluting stents[9]) and the use of antibodies that recognize epitopes specific to endothelial progenitor cells (e.g., anti-CD34-coated stents[12]). Both coating strategies have been designed to inhibit thrombosis mainly through promoting re-endothelialization of cardiovascular stents. Many clinical studies suggest that the Genous EPC-capture stent is a safe choice for patients with coronary disease[21-24]. However, Adrian et al.[25] reported that a similar late luminal loss of Genous EPC-capture stent to that of a bare-metal stent, despite initial optimism of rapid endothelialization.
In this study, our approach of surface modification has included the combination of VEGF and anti-CD34 antibody. The goal of this combination is to further accelerate endothelial repair, and thus further reduce the exposure time of stents in blood, decrease the rate of long-term thrombosis and shorten the time of antiplatelet therapy for patients. We firstly used dopamine-mediated heparin coating[13] and then a layer-by-layer method was employed to build multilayer films composed of heparin and VEGF on metal substrates. Based on the specific affinity of protein A and IgG antibodies, the protein A allows the subsequent immobilization of the anti-CD34 antibody. Our primary results of immunofluorescence and ELISA showed that the stainless metal steel was successfully coated with VEGF and anti-CD34 antibody (Figures 1 and 2). This research serves as a fundamental role for the novel coating strategy of simultaneous coating of VEGF and anti-CD34 antibody and further studies on the toxicity and effect of the combined coating are currently ongoing.
ACKNOWLEDGEMENTS
The study was supported by the following funds: National Natural Science Foundation of China through the National Outstanding Youth Science Fund (Grant number: 51103059); National Natural Science Foundation of Jilin Province (Grant numbers: 201115071, 20140101054JC); Jilin Industrial Technology Research and Development Project (Grant number: 2013C023-3); Science and Technology Development Planning Project of Jilin Province (Grant number: 20150519025JH); Scientific Research Planning Project of the Education Department of Jilin Province (Grant number: 2015).
REFERENCES
1. Carter AJ, Aggarwal M, Kopia GA, Tio F, Tsao PS, Kolata R, et al. Long-term effects of polymer-based, slow-release, sirolimus-eluting stents in a porcine coronary model. Cardiovasc Res. 2004;63(4):617-24. [MedLine]
2. Nakazawa G, Otsuka F, Nakano M, Vorpahl M, Yazdani SK, Ladich E, et al. The pathology of neoatherosclerosis in human coronary implants bare-metal and drug-eluting stents. J Am Coll Cardiol. 2011;57(11):1314-22. [MedLine]
3. Serruys PW, de Jaegere P, Kiemeneij F, Macaya C, Rutsch W, Heyndrickx G, et al. A comparison of balloon-expandable-stent implantation with balloon angioplasty in patients with coronary artery disease. Benestent Study Group. N Engl J Med. 1994;331(8):489-95. [MedLine]
4. Asai J, Takenaka H, Kusano KF, Ii M, Luedemann C, Curry C, et al. Topical sonic hedgehog gene therapy accelerates wound healing in diabetes by enhancing endothelial progenitor cell-mediated microvascular remodeling. Circulation. 2006;113(20):2413-24. [MedLine]
5. Ii M, Nishimura H, Iwakura A, Wecker A, Eaton E, Asahara T, et al. Endothelial progenitor cells are rapidly recruited to myocardium and mediate protective effect of ischemic preconditioning via "imported" nitric oxide synthase activity. Circulation. 2005;111(9):1114-20. [MedLine]
6. Finn AV, Joner M, Nakazawa G, Kolodgie F, Newell J, John MC, et al. Pathological correlates of late drug-eluting stent thrombosis: strut coverage as a marker of endothelialization. Circulation. 2007;115(18):2435-41. [MedLine]
7. Senger DR, Galli SJ, Dvorak AM, Perruzzi CA, Harvey VS, Dvorak HF. Tumor cells secrete a vascular permeability factor that promotes accumulation of ascites fluid. Science. 1983;219(4587):983-5. [MedLine]
8. Asahara T, Bauters C, Pastore C, Kearney M, Rossow S, Bunting S, et al. Local delivery of vascular endothelial growth factor accelerates reendothelialization and attenuates intimal hyperplasia in balloon-injured rat carotid artery. Circulation. 1995;91(11):2793-801. [MedLine]
9. Swanson N, Hogrefe K, Javed Q, Gershlick AH. In vitro evaluation of vascular endothelial growth factor (VEGF)-eluting stents. Int J Cardiol. 2003;92(2-3):247-51. [MedLine]
10. Lahtinen M, Blomberg P, Baliulis G, Carlsson F, Khamis H, Zemgulis V. In vivo h-VEGF165 gene transfer improves early endothelialisation and patency in synthetic vascular grafts. Eur J Cardiothorac Surg. 2007;31(3):383-90. [MedLine]
11. Lin HH, Chen YH, Yet SF, Chau LY. After vascular injury, heme oxygenase-1/carbon monoxide enhances re-endothelialization via promoting mobilization of circulating endothelial progenitor cells. J Thromb Haemost. 2009;7(8):1401-8. [MedLine]
12. Klomp M, Beijk MA, de Winter RJ. Genous endothelial progenitor cell-capturing stent system: a novel stent technology. Expert Rev Med Devices. 2009;6(4):365-75. [MedLine]
13. Bae IH, Park IK, Park DS, Lee H, Jeong MH. Thromboresistant and endothelialization effects of dopamine-mediated heparin coating on a stent material surface. J Mater Sci Mater Med. 2012;23(5):1259-69. [MedLine]
14. de Torre IG, Wolf F, Santos M, Rongen L, Alonso M, Jockenhoevel S, et al. Elastin-like recombinamer-covered stents: Towards a fully biocompatible and non-thrombogenic device for cardiovascular diseases. Acta Biomater. 2015;12:146-55. [MedLine]
15. Whitbourne RJ, Chamberlain AM, Hullihen DG, Rosebrough SF, Calistri-Yeh M. Medicated stent having multi-layer polymer coating. Google Patents; 2012.
16. Wright C, Llanos GH, Rakos R, King K, Falotico R. Methods and Devices for Delivering Therapeutic Agents to Target Vessels. Google Patents; 2012.
17. Gomes WJ, Buffolo E. Coronary stenting and inflammation. Rev Bras Cir Cardiovasc. 2003;18(4):III-VII. View article
18. Moses JW, Leon MB, Popma JJ, Fitzgerald PJ, Holmes DR, O'Shaughnessy C, et al.; SIRIUS Investigators. Sirolimus-eluting stents versus standard stents in patients with stenosis in a native coronary artery. N Engl J Med. 2003;349(14):1315-23. [MedLine]
19. Stone GW, Ellis SG, Cox DA, Hermiller J, O'Shaughnessy C, Mann JT, et al.; TAXUS-IV Investigators. A polymer-based, paclitaxel-eluting stent in patients with coronary artery disease. N Engl J Med. 2004;350(3):221-31. [MedLine]
20. Byrne RA, Sarafoff N, Kastrati A, Schömig A. Drug-eluting stents in percutaneous coronary intervention: a benefit-risk assessment. Drug Saf. 2009;32(9):749-70. [MedLine]
21. Lee YP, Tay E, Lee CH, Low A, Teo SG, Poh KK, et al. Endothelial progenitor cell capture stent implantation in patients with ST-segment elevation acute myocardial infarction: one year follow-up. EuroIntervention. 2010;5(6):698-702. [MedLine]
22. Chong E, Poh KK, Liang S, Lee RC, Low A, Teo SG, et al. Two-year clinical registry follow-up of endothelial progenitor cell capture stent versus sirolimus-eluting bioabsorbable polymer-coated stent versus bare metal stents in patients undergoing primary percutaneous coronary intervention for ST elevation myocardial infarction. J Interv Cardiol. 2010;23(2):101-8. [MedLine]
23. Aoki J, Serruys PW, van Beusekom H, Ong AT, McFadden EP, Sianos G, et al. Endothelial progenitor cell capture by stents coated with antibody against CD34: the HEALING-FIM (Healthy Endothelial Accelerated Lining Inhibits Neointimal Growth-First In Man) Registry. J Am Coll Cardiol. 2005;45(10):1574-9. [MedLine]
24. Duckers HJ, Soullie T, den Heijer P, Rensing B, de Winter RJ, Rau M, et al. Accelerated vascular repair following percutaneous coronary intervention by capture of endothelial progenitor cells promotes regression of neointimal growth at long term follow-up: final results of the Healing II trial using an endothelial progenitor cell capturing stent (Genous R stent). EuroIntervention. 2007;3(3):350-8. [MedLine]
25. Low AF, Lee CH, Teo SG, Chan MY, Tay E, Lee YP, et al. Effectiveness and safety of the genous endothelial progenitor cell-capture stent in acute ST-elevation myocardial infarction. Am J Cardiol. 2011;108(2):202-5. [MedLine]
Financial support: see in "Acknowledgements".
No conflict of interest.
Authors' roles & responsibilities
CLS: Analysis and interpretation of data; statistical analysis; design and study design; carried out operations and experiments
QL: Analysis and interpretation of data; statistical analysis; carried out operations and experiments
YPY: Analysis and interpretation of data; statistical analysis; carried out operations and experiments
GW: Analysis and interpretation of data; statistical analysis; carried out operations and experiments
JPW: Analysis and interpretation of data; statistical analysis; carried out operations and experiments
YL: Analysis and interpretation of data; statistical analysis; carried out operations and experiments
JCZ: Analysis and interpretation of data; statistical analysis; carried out operations and experiments
HYD: Analysis and interpretation of data; carried out operations and experiments
JGL: Analysis and interpretation of data; carried out operations and experiments
YHL: Analysis and interpretation of data; carried out operations and experiments
JL: Analysis and interpretation of data; carried out operations and experiments
YL: Analysis and interpretation of data; carried out operations and experiments
DC: Analysis and interpretation of data; carried out operations and experiments
BL: Analysis and interpretation of data; carried out operations and experiments
Article receive on Monday, August 18, 2014
 All scientific articles published at rbccv.org.br are licensed under a Creative Commons license
All scientific articles published at rbccv.org.br are licensed under a Creative Commons license