Ricardo Medeiros PiantáI; Andres Di Leoni FerrariII; Aline Almeida HeckIII; Débora Klein FerreiraIII; Jacqueline da Costa Escobar PiccoliIV; Luciano Cabral AlbuquerqueV; João Carlos Vieira da Costa GuaragnaVI; João Batista PetraccoVII
DOI: 10.5935/1678-9741.20140086
AVB: Atrioventricular block
CABG: Coronary artery bypass surgery
NYHA: New York Heart Association
PO: Postoperative period
POHS: Postoperative Heart Surgery Unit
PPM: Permanent pacemaker
PUCRS: Pontifical Catholic University of Rio Grande do Sul
TP: Temporary pacing
INTRODUCTION
Disturbances of the cardiac conduction system are relatively frequent in the postoperative period (PO) of coronary artery bypass grafting (CABG), with an incidence ranging from 18 to 55% of cases[1-6]. Atrioventricular block (AVB) is one of these conduction disturbances and its incidence ranges from 0.5 to 16%[3,5-9]. Most patients have transitory and reversible conduction disorders that require temporary pacing (TP). 0.4 to 1.1% of patients, however, when faced with the irreversibility of the condition, will have to undergo a permanent pacemaker (PPM) implant during their hospital stay[10]. This study, which is unprecedented in the national literature, tries to identify the relationship between pre-, intra and postoperative (perioperative) factors associated with the emergence of AVB, the need for TP and, if the case, the implantation of a PPM in the PO of CABG.
METHODS
Population and sample
Between January 1996 and December 2012, 3532 CABGs with cardiopulmonary bypass (CPB) were performed at the Sao Lucas Hospital of the Pontifical Catholic University of Rio Grande do Sul (PUCRS).
Study Design
Historical cohort observation study. Data were collected prospectively and entered into the database of the Postoperative Heart Surgery Unit (POHS) of the São Lucas Hospital of PUCRS.
Inclusion Criteria
Patients with age equal to or greater than 18 years who were submitted to isolated CABG.
Exclusion Criteria
Patients who had been undergo valvular surgery, for left ventricular aneurysmectomy or correction of the interventricular communication associated with CABG.
Study Variables
Age - the mean age was calculated and also divided into groups for analysis: less than 60 years and greater than or equal to 60 years, according to the reference in the literature[11,12]; gender (male and female); left ventricular ejection fraction (EF) - evaluated by echocardiography or radiocardiography, with the values being subdivided for analysis into < 40% and > 40 %; chronic kidney disease (CKD) - diagnosed through serum creatinine level > 1.5 mg/dl, according to the reference in the literature[11,12]; Diabetes Mellitus (DM); atrial fibrillation (AF); previous use of beta-blockers; previous use of statins; previous use of other antiarrhythmics (propafenone and/or amiodarone); acute myocardial infarction (AMI) prior to CABG; New York Heart Association functional (NYHA) class; presence of calcification of the aorta; time of cardiopulmonary bypass (CPB); aortic clamping time; need for the use of intra-aortic balloon (IAB) in the PO of CABG; perioperative AMI; length of hospital stay and hospital death.
Outcome
Development of AVB in the PO of CABG and the need for TP and implantation of a PPM.
Procedures
The CABGs were performed under general anesthesia. In all cases, a hyperkalemic cardiac arrest was induced using a cold cardioplegic blood solution in the anterograde flow, with the infusion being repeated every 20 minutes. A mild systemic hypothermia (32ºC) was used. After surgery, all patients were transferred to the ICU of the POHS with mechanical ventilation.
Statistical Analysis
The data was plotted in a digital Microsoft Access® spreadsheet and analyzed using version 17.0 of the statistical software SPSS. The descriptive analysis was performed through frequency and mean ± standard deviation analysis, according to the case. For the univariate analysis the following tests were performed: chi-square and/or Fisher's Exact Test for ordinal variables and the Student's T-test for quantitative data. The multivariate analysis was performed using logistic regression (backward conditional method). The difference was considered as statistically significant for the value of P<0.05.
Ethical Considerations
The design of this study was submitted to the Research Ethics Committee of the Faculty of Medicine of the PUCRS, under registration number 060/3478.
RESULTS
Of the 3532 patients undergoing CABG in the period under analysis, 288 (8.15%) presented the clinical and electrocardiographic signs of AVB during the postoperative period, with an indication for temporary pacing (TP).
Table 1 shows the demographic profile of the patients studied. The univariate analysis of the preoperative data revealed a greater need for TP in the PO of CABG in patients above 60 years of age (OR=2.48; CI 95% 1.90-3.24; P<0.0001), of the female gender (OR=1.03; CI 95% 1.00-1.05; P=0.012), with CKD (OR=2.03; CI 95% 1.55-2.65; P<0.0001), the presence of AF (OR=2.38; CI 95% 1.49-3.72; P<0.0001) and in patients with NYHA functional class III or IV (OR=1.60; CI 95% 1.21-2.12; P=0.001).
Table 2 presents the trans and postoperative data, together with their univariate analysis. Here we can observe the association of AVB with the need of TP in patients who presented calcification of the aorta, perioperative AMI and the need for the use of an IAB. Of statistically significant relevance, the univariate analysis also revealed the association of TP caused by AVB with increased mortality (17.9% vs. 7.3%) and with a longer hospital stay (mean hospitalization time of 12.75 days compared to 10.53 days for those who did not require TP).
These data were submitted to multivariate analysis (Table 3), which revealed a higher risk of AVB in the PO of CABG in patients with: age > 60 years, female sex, CKD, AF, NYHA functional class III or IV, perioperative AMI and with the use of an IAB. Patients with EF<40 %, DM, the use of beta-blockers, statins and other antiarrhythmic drugs, prior AMI and CPB and aortic clamping times didn't prove to be independent risk variables for the development of AVB in the PO of CABG.
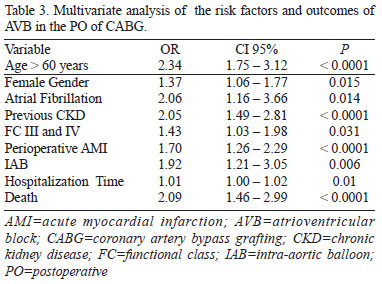
In the multivariate analysis, the presence of AVB resulted in a longer hospital stay (12.75 days vs. 10.53 days for those who didn't develop AVB) (OR=1.01; CI 95% 1.00-1.02; P=0.01) and in a significant increase in the risk of mortality (17.9% vs. 7.3% for patients without AVB) (OR=2.09; CI 95% 1.46-2.99; P<0.0001).
In the subgroup of 288 patients who had AVB and who had undergone TP, 08 (2.78 %) required a PPM implant, corresponding to 0.23% of the total cohort analyzed. The average time elapsed since the surgery until the PPM implant was 12.25 days.
DISCUSSION
CABG is a proven therapeutic strategy for the treatment of Coronary Artery Disease (CAD). Although it is well tolerated by most patients, perioperative complications can occur, among which we find disturbances in the cardiac conduction system in varying degrees, including AVB.
Previous studies have reported an incidence of conduction disturbances (CD) after CABG that varies from 18 to 55% of cases[1-6], with the right bundle branch block being the most common[4]. Atrioventricular block (AVB) is one of these conduction disturbances and its incidence ranges from 0.5 to 16%[3,5-9]. Our incidence of AVB is in line with this data, since 8.15% of our patients developed AVB in the PO of CABG.
The etiology of AVB seems to be multifactorial. The patient's age (>60 years), hypertension, number of revascularized vessels, aortic clamping time, total time of CPB, use of digitalis and beta-blockers, type of cardioplegia and previously existing left bundle branch block may be related to its appearance[1-4,8,9,13,14].
Myocardial ischemia seems to be the factor that is most implicated in the emergence of AVB, since there is a correlation with coronary artery disease (CAD) and preoperative AMI[4]. Studies[3,10] have demonstrated that perioperative AMI also increases the incidence of AVB in the PO of CABG. Caspi et al.[7] reported a higher occurrence of AVB in patients with AMI in the PO of CABG (12% vs. 2 %, P<0.05).
However, AMI before CABG was not a significant factor for the appearance of AVB in our study, which is consistent with the world literature[3,7,8,15,16]. This shows there is no difference in the incidence of AVB between patients who had preoperative AMI and those who didn't, regardless of its electrocardiographic location.
Caspi et al.[7] have shown that the combination of left main disease and proximal obstruction of a dominant right coronary artery was more frequent in patients who exhibited AVB (32%) than in those who without it (12%, P<0.05). The explanation for this effect is the fact that the cardioplegic solution is not properly distributed to the coronary beds because of their high degree of obstruction, which compromises myocardial protection and, in some cases, because of the impossibility of bypassing the right coronary artery.
The impairment of myocardial irrigation gets worse with age, just as the frequency of degenerative diseases of the conduction system, increasing the probability of AVB[9,11,15,17-19]. In this scenario, our patients above 60 years of age presented a significant risk (OR=2.34; CI 95% 1.75-3.12; P<0.0001) for the development of AVB in the PO of CABG, corroborating the findings of other studies[7,8,13,14].
The electrical cardiac conduction tissue differs from cardiac myocytes by being less tolerant to the effects of ischemia, hyperkalemia and hypothermia (whether these are systemic or, mostly, induced by a cardioplegic solution that is cold and rich in potassium). This may cause a transient block of the conduction system[11]. The advent of cold cardioplegia as a method of myocardial protection has increased the incidence of CD from 20 to 58%[13]. The more significant incidence of conduction disturbances occurred in patients who received cold cardioplegia, as opposed to warm (19.6% vs. 1.7 %, respectively)[13], a finding that has also been described by Sirlak et al.[20]. Specifically with respect to AVB, the incidence was of 3.8% in the hypothermia group and zero in the normothermal group[13]. All patients in our study underwent surgery with the myocardial protection performed by infusion of a cold cardioplegic blood solution at the root of the aorta every 20 minutes, which contributed to the genesis of AVB cases.
As such, the perfusion injury determined by the myocardial ischemia and the hypothermic injury caused by the cardioplegic solution are the mechanisms that are most involved in the genesis of AVB, acting on the proximal portions of the bundle of His, which are more sensitive to this type of aggression than the more distal conduction tissue, determining the emergence of bundle branch blocks and increasing the risk of AVB[4].
In this scenario, the extent of the CAD, the duration of CPB and the aortic clamping time could compromise myocardial protection during surgery, increasing the risk of an ischemic injury and of metabolic damage to the conduction tissue[11]. However, our CPB time of > 90 min and aortic clamping time of > 40 min showed no influence on the development of AVB, which is supported by the literature[5-7,16,19]. Baerman et al.[1], however, demonstrated that patients with lower CPB (101±32min x 121±34min; P<0,01) and aortic clamping (44±19min x 53±17min; P<0.05) times didn't show evidence of AVB in the PO of CABG.
Our study has shown that the female gender is a risk factor for the occurrence of AVB (OR=1.37, CI 95% 1.06-1.77; P=0.015), which contrasts with the results of Gordon et al.[19] who observed a higher need for PPM implants in men (P=0.041). Other studies[3,8,15,20], however, didn't point to any of the genders as risk factor. Cadore et al.[12] had already pointed to the female gender as a risk predictor for mortality in CABG, which can be an expression of the greater severity of the ischemic impairment in this gender and explain their greater tendency for developing the block, as seen in our study.
The presence of CKD was also verified to be a risk factor for the development of AVB (OR = 2.05; CI 95% 1.49-2.81; P<0.0001). A previous study[19] indicated the presence of CKD as more significant among those patients who required PPM implantation in the PO of CABG. Like the female gender variable, CKD was also found to be a predictor of mortality in patients underwent CABG according to the score by Cadore et al.[12], expressing its potential for increasing the risk of complications in the PO of CABG.
Another risk predictor for the occurrence of AVB was the more advanced functional class of the NYHA (III and IV) (OR = 1.43; CI 95% 1.03-1.98; P=0.031). Studies[18,19] have corroborated this finding, indicating that patients who underwent heart surgery and needing a PPM implant were in the more severe functional class of the NYHA (III and IV) when compared to patients who did not require such an implant (57% vs. 35%, respectively, P<0.0001)[18]. Bateman et al.[6] showed that of those patients who passed away within the first 30 days of the PO of CABG and who had developed some degree of blockage, 90% were into class IV of the NYHA in the preoperative period.
The patients in this study who had EF<40% did not present a significant risk for the appearance of AVB in the PO of CABG (OR=1.11; CI 95% 0.85-1.45; P=0.44), a finding supported by Gordon et al.[19] who didn't observe any significant impact of EF on the need for PPM implantation in the PO of isolated CABG. Caspi et al.[7], however, found a greater susceptibility to the development of atrioventricular block in patients submitted to CABG with a lower EF.
Although Merin et al.[18] mention that the use of antiarrhythmic agents is more frequent in the group of patients that develops blockage after heart surgery (CABG, valve or combined), our data does not reflect this influence. Regarding the use of beta-blockers, we also didn't find any association with the development of AVB, which has already been described by other authors[2,3,16].
The need for the use of an IAB in the PO of CABG occurred in 141 patients (4%) of the total sample of 3532 patients, of which 20.6% developed AVB, leading to the need for TP (OR=1.92; CI 95% 1.21-3.05; P=0.006). The need for the use of the IAB has been associated with a greater probability of developing blocking and has been indicated as a predictor of its occurrence and of the need for a PPM implant[6,17,19]. Probably because its use is an expression of a more significant ischemic cardiopathy, i.e., of patients with more severe compromising. This finding is important because the patients did not have AVB in the preoperative period, presumably reflecting a greater perioperative myocardial injury[6].
Perioperative AMI was a risk factor for the emergence of AVB (OR=1.70; CI 95% 1.26-2.29; P<0.0001), and this was corroborated by the study of Caspi et al.[7] who identified the occurrence of low cardiac output (34% vs. 3%) and perioperative AMI (12% x 2%) as risk factors for AVB in the PO of CABG. Perioperative AMI also increases the need for a PPM implant[10], reflecting acute ischemic damage of the conduction tissue.
The need for TP showed a significant association with mortality (OR=2.09; CI 95% 1.46-2.99; P<0.0001), which was 17.7% for patients with AVB and 7.2% for those who didn't develop it. Zeldis et al.[3] had already reported a mortality of 19.2% in the group of patients who developed block of the left conduction system (left bundle branch block or left anterior hemiblock or both), compared with a 7% mortality rate in the group of patients without such block. Specifically with respect to AVB, Caspi et al.[7] observed a significantly higher mortality in the group of patients who developed AVB (7% vs. 0.6%). On the other hand, patients who develop right bundle branch block or fascicular block have a more favorable prognosis, because these are more transient disorders and because they do not increase mortality[6,21].
The patients who developed AVB had a significantly longer hospital stay (mean hospitalization time of 12.75 days compared to 10.53 days for those who did not need TP for AVB (OR=1.01 CI 95% 1.00-1.02; P=0.01). Gordon et al.[19] have shown that the need for a PPM implant significantly increased hospital stay (23.3±18.7 days vs. 9.6±9.0 days for patients without need of implant, P=0.0001) and ICU stay (5.6±10.5 days vs. 2.2±3.3 days, P=0.0258). Other studies[11,18] also corroborate this finding of a longer hospital stay in the presence of AVB and the need of TP.
In our study, the need for a PPM implant occurred in 08 of the 3532 patients studied (0.23%), which is lower than the rate found in the literature, which points to the need for PPM implants in 0.49% of AVB cases[8]. Gordon et al.[19] implanted PPMs in 50 of their 6859 patients submitted to CABG (0.73%). When other types of post-CABG conduction blocks are considered, the incidence of implants rises and ranges from 0.4 to 1.1%[10]. The calculated risk for need of a PPM implant in the PO of non-complicated CABG is 0.9%[19].
Nascimento et al.[22] couldn't identify any prognosis criterion for the reversibility of AVB in the PO of heart surgery. The ideal moment for the implantation of a PPM in the PO of CABG hasn't yet been properly established. According to the Brazilian Guidelines for Implantable Electronic Heart Devices[23], patients with asymptomatic AVB with wide QRS after cardiac surgery that persists after 15 days, are indicated for a PPM implantation (Class I, level of evidence C). In the cases of asymptomatic AVB persisting after 15 days, resulting from cardiac surgery, with narrow QRS or nodal escape rhythm and good chronotropic response, and in those cases without the prospect of reversal (< 15 days) PPM implantation is also indicated (Class IIa, level C).
According to the criteria of the American College of Cardiology and the American Heart Association, a PPM implant is indicated in 3rd and advanced 2nd degree AVB in the postoperative period of heart surgery, in addition to cases without expectation of resolution. The decision regarding the time of the implant should be taken by the physician[24].
The European Society of Cardiology recommends a waiting period of 5 to 7 days for the resolution of transient bradyarrhythmias after cardiac surgery, before the decision for the implant is made[25].
According to Pires et al.[13] and Merin et al.[18], the decision to perform the implant should be taken between the 4th and 5th day of the PO, because if the AVB or dysfunction of the sinus node are still present up to this moment, then they tend to be permanent. This would facilitate the early mobilization of patients and shorten their hospitalization time.
Of the 288 patients in our study who had AVB, 08 received a PPM implant after an average of 12.25 days into the PO, which is in line with the Brazilian (Class IIa, level of evidence C), American and European (Class I, level C) guidelines. Emlein et al.[8] described a series of 8 patients who underwent a PPM implant after developing AVB with an average of 10.5±6.5 days into the PO.
Limitations of the Study
The limitations of this study are those inherent to a retrospective database analysis, but they reflect the significant years of experience of an academic institution. Within these limitations we can cite the relative difficulty of accessing the full data, which causes a potential risk of not measuring some random variables. The fact that the results come from the sample of a single center can also represent some degree of bias in the treatment. Another limitation of this study is the absence of more precise information regarding the height of the atrioventricular conduction disorder and the existence or not of any escape rhythm.
Regarding the PPM implants performed in our study, they followed the recommendations of Brazilian, American and European guidelines almost strictly. In this small group of patients a more thorough analysis was compromised, but this could be the target of a more detailed study to be developed in the future.
CONCLUSION
This work sheds light on the risk factors associated with the development of AVB in the PO of CABG and the consequent need for TP and a definitive pacemaker. Based on this we could establish that female patients, 60 years of age or more, with the diagnosis of AF and CKD, in stages III and IV of the functional class, who had perioperative AMI and required the use of an IAB, have a higher risk of developing AVB in the PO of CABG. AVB determines a more prolonged hospitalization and, what is more important, doubles the risk of mortality.
REFERENCES
1. Baerman JM, Kirsh MM, Buitleir M, Hyatt L, Juni JE, Pitt B, et al. Natural history and determinants of conduction defects following coronary artery bypass surgery. Ann Thorac Surg. 1987;44(2):150-3. [MedLine]
2. Wexelman W, Lichstein E, Cunningham JN, Hollander G, Greengart A, Shani J. Etiology and clinical significance of new fascicular conduction defects following coronary bypass surgery. Am Heart J. 1986;111(5):923-7. [MedLine]
3. Zeldis SM, Morganroth J, Horowitz LN, Michelson EL, Josephson ME, Lozner EC, et al. Fascicular conduction disturbances after coronary bypass surgery. Am J Cardiol. 1978;41(5):860-4. [MedLine]
4. Kumbhani DJ, Sharma GV, Khuri SF, Kirdar JA. Fascicular conduction disturbances after coronary artery bypass surgery: a review with a meta-analysis of their long-term significance. J Card Surg. 2006;21(4):428-34. [MedLine]
5. Kirdar JA, Sharma GV, Khuri SF, Josa M, Parisi AF. Pathogenesis and prognostic significance of conduction abnormalities after coronary bypass surgery. Cardiovasc Surg. 1996;4(6):832-6. [MedLine]
6. Bateman TM, Weiss MH, Czer LS, Conklin CM, Kass RM, Stewart ME, et al. Fascicular conduction disturbances and ischemic heart disease: adverse prognosis despite coronary revascularization. J Am Coll Cardiol. 1985;5(3):632-9. [MedLine]
7. Caspi J, Amar R, Elami A, Safadi T, Merin G. Frequency and significance of complete atrioventricular block after coronary artery bypass grafting. Am J Cardiol. 1989;63(9):526-9. [MedLine]
8. Emlein G, Huang SK, Pires LA, Rofino K, Okike ON, Vander Salm TJ. Prolonged bradyarrhythmias after isolated coronary artery bypass graft surgery. Am Heart J. 1993;126(5):1084-90. [MedLine]
9. Rocha AS, Pitella FJ, Lorenzo AR, Barzan V, Colafranceschi AS, Brito JO, et al. Age influences outcomes in 70-year or older patients undergoing isolated coronary artery bypass graft surgery. Rev Bras Cir Cardiovasc. 2012;27(1):45-51. [MedLine] View article
10. Emkanjoo Z, Mirza-Ali M, Alizadeh A, Hosseini S, Jorat MV, Nikoo MH, et al. Predictors and frequency of conduction disturbances after open-heart surgery. Indian Pacing Electrophysiol J. 2008;8(1):14-21. [MedLine]
11. Ferrari AD, Süssenbach CP, Guaragna JC, Piccoli JC, Gazzoni GF, Ferreira DK, et al. Atrioventricular block in the postoperative period of heart valve surgery: incidence, risk factors and hospital evolution. Rev Bras Cir Cardiovasc. 2011;26(3):364-72. [MedLine] View article
12. Cadore MP, Guaragna JC, Anacker JF, Albuquerque LC, Bodanese LC, Piccoli Jda C, et al. A score proposal to evaluate surgical risk in patients submitted to myocardial revascularization surgery. Rev Bras Cir Cardiovasc. 2010;25(4):447-56. [MedLine] View article
13. Pires LA, Wagshal AB, Lancey R, Huang SK. Arrhythmias and conduction disturbances after coronary artery bypass graft surgery: epidemiology, management, and prognosis. Am Heart J. 1995;129(4):799-808. [MedLine]
14. Baraka AS, Taha SK, Yazbeck VK, Rizkallah PA, Zughbi JP, Aouad MJ, et al. Transient atrioventricular block after release of aortic cross-clamp. Anesth Analg. 1995;80(1):54-7. [MedLine]
15. Mosseri M, Meir G, Lotan C, Hasin Y, Applebaum A, Rosenheck S, et al. Coronary pathology predicts conduction disturbances after coronary artery bypass grafting. Ann Thorac Surg. 1991;51(2):248-52. [MedLine]
16. Hippeläinen M, Mustonen P, Manninen H, Rehnberg S. Predictors of conduction disturbances after coronary bypass grafting. Ann Thorac Surg. 1994;57(5):1284-7.
17. Cook DJ, Bailon JM, Douglas TT, Henke KD, Westberg JR, Shirk-Marienau ME, et al. Changing incidence, type, and natural history of conduction defects after coronary artery bypass grafting. Ann Thorac Surg. 2005;80(5):1732-7. [MedLine]
18. Merin O, Ilan M, Oren A, Fink D, Deeb M, Bitran D, et al. Permanent pacemaker implantation following cardiac surgery: indications and long-term follow-up. Pacing Clin Electrophysiol. 2009;32(1):7-12. [MedLine]
19. Gordon RS, Ivanov J, Cohen G, Ralph-Edwards AL. Permanent cardiac pacing after a cardiac pperation: predicting the use of permanent pacemakers. Ann Thorac Surg. 1998;66(5):1698-704. [MedLine]
20. Sirlak M, Eryilmaz S, Yazicioglu L, Kiziltepe U, Inan MB, Taşöz R, et al. Conduction disturbances in coronary artery bypass surgery. Int J Cardiol. 2003;92(1):43-8. [MedLine]
21. Vogler J, Breithardt G, Eckardt L. Bradiarritmias y bloqueos de laconducción. Rev Esp Cardiol. 2012;65(7):656-67.
22. Nascimento CS, Viotti Junior LA, Silva LHF, Araújo AM, Bragalha AMLA, Gubolino LA. Bloqueio atrioventricular de alto grau induzido pela cirurgia cardíaca: estudo de critérios de reversibilidade. Rev Bras Cir Cardiovasc. 1997;12(1):56-61. View article
23. Martinelli Filho M, Zimerman LI, Lorga AM, Vasconcelos JTM, Lorga Filho A, Fagundes AA, et al. Guidelines for Implantable Electronic Cardiac Devices of the Brazilian Society of Cardiology. Arq Bras Cardiol. 2007;89(6):e210-38.
24. Epstein AE, DiMarco JP, Ellenbogen KA, Estes NA 3rd, Freedman RA, Gettes LS, et al; American College of Cardiology Foundation; American Heart Association Task Force on Practice Guidelines; Heart Rhythm Society. 2012 ACCF/AHA/HRS focused update incorporated into the ACCF/AHA/HRS 2008 guidelines for device-based therapy of cardiac rhythm abnormalities: a report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines and the Heart Rhythm Society. J Am Coll Cardiol. 2013;61(3):e6-75. [MedLine]
25. European Society of Cardiology (ESC); European Heart Rhythm Association (EHRA), Brignole M, Auricchio A, Baron-Esquivias G, Bordachar P, Boriani G, Breithardt OA, et al. 2013 ESC guidelines on cardiac pacing and cardiac resynchronization therapy: the task force on cardiac pacing and resynchronization therapy of the European Society of Cardiology (ESC). Developed in collaboration with the European Heart Rhythm Association (EHRA). Europace. 2013;15(8):1070-118. [MedLine]
No financial support.
Authors' roles & responsibilities
RMP: Analysis and/or interpretation of data; final approval of the manuscript; study design; conduct of operations and/or experiments; writing of the manuscript or critical review of its content
ADLF: Analysis and/or interpretation of data; study design; conduct of operations and/or experiments; writing of the manuscript or critical review of its content
AAH: Analysis and/or interpretation of data; actual operations and/or experiments; writing of the manuscript or critical review of its content
DKF: Analysis and/or interpretation of the data
JCEP: Analysis and/or interpretation of the data; statistical analysis
LCA: Final approval of the manuscript; conduct of operations and/or experiments; writing of the manuscript or critical review of its content
JCVCG: Final approval of the manuscript; writing of the manuscript or critical review of its content
JBP: Conduct of operations and/or experiments; writing of the manuscript or critical review of its content
Article receive on Monday, December 16, 2013
 All scientific articles published at rbccv.org.br are licensed under a Creative Commons license
All scientific articles published at rbccv.org.br are licensed under a Creative Commons license