ABSTRACT
Pulmonary artery coarctation often happens in patients with pulmonary atresia. The correction has been usually performed using cardiopulmonary bypass and during the neonatal period, influencing pulmonary artery development and prognosis. Three patients with pulmonary atresia with PDA underwent correction using median sternotomy without cardiopulmonary bypass. The PDA maintained the arterial saturation during Blalock Taussig anastomoses upon the contralateral pulmonary artery. Arterioplasty was performed using an autologous pericardium with 7-0 PDS running suture and saturation was maintained by Blalock shunt. All patients presented good follow-up and where discharged with good enlargment of coarctation area.
RESUMO
A coarctação de artéria pulmonar é comum em pacientes com atresia pulmonar. A correção tem sido com CEC e no período neonatal, quando influencia o desenvolvimento das artérias pulmonares e o prognóstico. Foram corrigidos três pacientes por esternotomia mediana com atresia pulmonar dependentes do ducto arterioso (PCA) sem uso de CEC. O PCA mantinha a saturação durante a confecção do Blalock Taussig na artéria pulmonar contralateral. Arterioplastia foi realizada com sutura de pericárdio autólogo com PDS 7-0 e saturação mantida pelo Blalock. Todos pacientes tiveram boa evolução e alta hospitalar com avaliação de controle demonstrando bom alargamento da área coarctada.
INTRODUCTION
The pulmonary artery stenosis at the insertion site of the ductus arteriosus, also known as coarctation of the pulmonary artery was described in 1954 by Sondergaard [1]. The pulmonary artery stenosis justaductal is extremely common and can occur in up to 60% of cases with pulmonary atresia and up to 10% of patients with pulmonary stenosis [2-4]. Oliveira et al. [5] described in our country, in a study of 766 catheterizations assessment that there was pulmonary obstruction in 37.7% of cases with pulmonary atresia, 8.6% in cases with pulmonary stenosis, 17.2% in cases of tetralogy of Fallot and 28.3% in cases of right ventriculcar double outflow tract with pulmonary stenosis. In cases of pulmonary atresia with atresia of the pulmonary trunk, the levels of obstruction can reach 75% [2].
Surgical correction of coarctation of the pulmonary artery should be performed as early as possible, especially in patients who have indication for univentricular correction [6]. The completion of correction of coarctation of the pulmonary artery allows proper development of the pulmonary tree, favoring the vascularization and maintenance of low resistance in the pulmonary arteries [7]. With the delay of surgical treatment, future procedures may be more complex and with less favorable outcomes due to the underdevelopment of the lung, which receives less blood flow [8].
Currently, the treatment of coarctation of the pulmonary artery has been accomplished even in the neonatal period and with use of cardiopulmonary bypass in several centers around the world [6,7,9]. The use of cardiopulmonary bypass in the neonatal period causes to the child a large inflammatory stimulation and can double its intravascular blood volume [10]. Furthermore, it may be necessary to use more severe hypothermia, in order to decrease the arterial flow to facilitate the visualization of the operative field, with the possibility of neurological deficit [11].
Therefore, the aim of this study was to demonstrate the feasibility and immediate results of surgical correction of coarctation of the pulmonary artery without cardiopulmonary bypass, as previously reported by Oliveira et al. [12].
METHODS
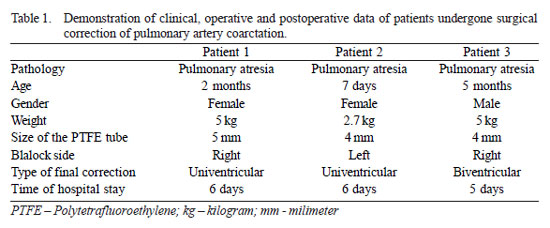
In the period between April and October 2008, three children with coarctation of the pulmonary artery (CoAPA) underwent surgery at the National Institute of Cardiology without cardiopulmonary bypass. Two children were female and one male. The weight of one patient was 2.7 kg and 5 kg of the other two. One child was 7 days old and the other two 2 and 4 months (Table 1).
Two children presented left justaductal pulmonary stenosis and one right with a ductus arteriosus nourishing the right branch of the pulmonary artery. Two presented pulmonary atresia (PA) with interventricular communication (IC), but with hypoplastic right ventricle, and one, pulmonary atresia with IC with possibility of biventricular treatment, but presented in cyanotic crisis indicating confection of systemic-pulmonary shunt. All children were diagnosed with multidetector computed tomography angiography prior to surgery and underwent examination of control in the postoperative period.
Surgical technique
The procedure was performed with monitoring of invasive pressure in the radial artery contralateral to the subclavian artery used for confection of the systemic-pulmonary shunt. We used deep venous approach with double lumen catheter and indwelling catheter. The surgical approach used in all children was median sternotomy, with partial resection of the thymus tissue.
The pericardial sac was incised in an inverted T manner, with removal of a piece of autologous pericardium, kept in saline solution for reconstruction of the pulmonary artery. A wide dissection of the great vessels and patent ductus arteriosus (PDA) was performed, exposing clearly the region of coarctation of the pulmonary artery (Figure 1). All children had the PDA dissected carefully to maintain the arterial oxygen saturation during the consfection of systemic-pulmonary shunt on the contralateral pulmonary artery.

Fig. 1 - Anatomical view of coarctation of the pulmonary artery and its relationship with the patent ductus arteriosus (PDA). The aortic artery is seen posteriorly to the pulmonary artery only for illustration purposes. The aortic and pulmonary vessels are carefully dissected, avoiding injury or spasm of the PDA, which is used to maintain arterial oxygen saturation
It was used infusion of a bolus of 1 mg/kg of heparin, which was peaked if the activated clotting time (ACT) was less than 250 seconds. The pulmonary artery contralateral to the PDA was widely dissected and fixed by yarns. The remnant of the pulmonary artery was ligated and sectioned to prevent distortion of the pulmonary artery branches. The subclavian artery, near the brachiocephalic trunk was dissected and fixed using yarns, receiving then an atraumatic vascular clamp in the lateral position. It was performed a longitudinal arteriotomy of ideal size for anastomosis of the polytetrafluoroethylene (PTFE) tube, previously cut in bevel. The PTFE tube of 4 or 5 mm was anastomosed, end-to-side, on the subclavian artery, using continuous suture with 7-0 polypropylene yarn (Figure 1B). The pulmonary artery received then two straight atraumatic vascular clamps in transverse position, allowing no distortion of the PDA. The systemic-pulmonary shunt was completed with end-to-side implantation of the PTFE tube into the pulmonary artery with continuous suture using 7-0 polypropylene.
During the entire procedure of confection of the systemic-pulmonary shunt, the arterial saturation was maintained by the flow of blood through the PDA at the contralateral pulmonary artery (Figure 2). The clamps of the subclavian and pulmonary arteries were removed, allowing flow through the systemic-pulmonary shunt, which maintained the blood oxygen saturation during the reconstruction of the coarcted pulmonary artery.
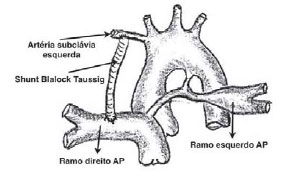
Fig, 2- Confection of systemic-pulmonary shunt, modified Blalock Taussig, of the subclavian artery up to the pulmonary artery contralateral to the PDA with polytetrafluoroethylene (PTFE) tube of 4 or 5 millimeters anastomosed with a continuous suture using 7-0 polypropylene yarn
The PDA was then widely dissected and connected twice with 6-0 polypropylene suture. Then, the PDA was sectioned, allowing wide dissection of the remainder of the contralateral pulmonary artery to the systemic-pulmonary shunt. We used two straight atraumatic vascular clamps, so as to leave wide exposure of the pulmonary artery, which includes the segment of narrowing of the coarctation. During this period, the blood oxygen saturation was maintained by the flow of blood through the systemic-pulmonary shunt, previously confectioned. It was performed a wide longitudinal opening in the pulmonary artery, including the segment of coarctation. The piece of fresh autologous pericardium was used for the enlargement of the pulmonary artery using continuous suture with absorbable polydioxanone (PDS) 7-0 yarn (Figure 3). After the angioplasty with autologous pericardium, the atraumatic vascular clamps were removed, restoring complete blood flow for both pulmonary arteries by the influx of blood through the systemic-pulmonary shunt. Heparinization was maintained by infusion pump for at least 24 hours to avoid the formation of thrombi in segments manipulated.

Fig. 3 - View of wide longitudinal enlargement of the area of coarctation of the pulmonary artery using a graft of fresh autologous pericardium with a continuous suture of absorbable 7- 0 polydioxanone (PDS) yarn. The PDA is ligated and sectioned between double ligature with 6-0 polypropylene, to prevent distortion of the pulmonary branches
Two PTFE tubes of 4 mm and 5 mm were implanted, using the technique described above. All surgeries were performed with excellent hemodynamic stability and without cardiopulmonary bypass. All patients presented good performance and discharge from hospital, one with 5 and two with 6 days postoperatively (Table 1). All patients underwent control assessment with good enlargement of the CoAPA region assessed by multidetector computed tomography angiography (Figure 4). The children present no bleeding due to maintenance of heparin.

Fig. 4 - View of multidetector CT angiography in patients undergoing surgical correction of coarctation of the pulmonary artery
Coarctation of the pulmonary artery is a rare disease first described more than 50 years ago and very common in cases with some degree of stenosis of the right ventricular outflow tract [1-3]. Luhmer et al. [13] has shown that 36%, nine out of 25 neonates with cyanosis due to obstruction of the right ventricular outflow tract, presented justaductal pulmonary stenosis associated with the invasion of ductal tissue in the pulmonary artery. Due to lack of knowledge and/or investigation of obstructions at the level of the pulmonary arteries, many children have been only diagnosed only later, worsening the prognosis, especially in cases progressing to the Fontan operation.
The need for early surgery, especially in cases with indication of univentricular repair is mandatory and advocated by several centers around the world [6,7,9,14]. The main objective is to restore adequate flow to both pulmonary arteries. Thus, it may prevent unilateral hypoplasia of the pulmonary arterial tree and therefore the growth of tortuous bronchial collateral arteries [13].
Surgical correction with enlargement of the coarctation area using autologous pericardium is performed by several groups [6,7,9]. The surgery is usually performed with cardiopulmonary bypass, which leads to an exacerbation of the inflammatory response with increased hospital stay and complications [10]. The period of hospital stay in this group of three patients was, at maximum, of 6 days (Table 1) extremely short when compared with an average of 13.5 ± 12.3 days, only of stay in the ICU, reported by other groups that perform the procedure with cardiopulmonary bypass [9].
The possibility of confection of the systemic-pulmonary shunt using the PDA to maintain arterial oxygen saturation is essential for performing the procedure without cardiopulmonary bypass [12]. For this, one should dissect the anatomical structures without the occurrence of injuries, spasms or distortion of the PDA during the preparation and anastomoses of the systemic-pulmonary shunt (Figure 1C).
CONCLUSIONS
Even in a group with a small number of patients, this tactic seems to be favorable for the treatment of coarctation of the pulmonary artery, avoiding the use of cardiopulmonary bypass and its deleterious effects. The accomplishment of surgery in the neonatal period provides smooth enlargement of the pulmonary artery stenosis with the possibility of good development, especially in the field of candidates for Fontan operation.
REFERENCES
1. Sondergaard T. Coarctation of the pulmonary artery. Dan Med Bull. 1954;1(2):46-8. [MedLine]
2. Elzenga NJ, Gittenberger-de Groot AC. The ductus arteriosus and stenoses of the pulmonary arteries in pulmonary atresia. Int J Cardiol. 1986;11(2):195-208. [MedLine]
3. Elzenga NJ, von Suylen RJ, Frohn-Mulder I, Essed CE, Bos E, Quaegebeur JM. Juxtaductal pulmonary artery coarctation. An underestimated cause of branch pulmonary artery stenosis in patients with pulmonary atresia or stenosis and a ventricular septal defect. J Thorac Cardiovasc Surg. 1990;100(3):416-24. [MedLine]
4. Momma K, Takao A, Ando M, Nakazawa M, Satomi G, Imai Y, et al. Juxtaductal left pulmonary artery obstruction in pulmonary atresia. Br Heart J. 1986;55(1):39-44. [MedLine]
5. Oliveira DM, Monteiro A, Simões LCN, Colafranceschi AS. Fatores anatômicos de risco para o desenvolvimento de obstruções nas artérias pulmonares. Rev SOCERJ. 2009;21(supl A):32.
6. Ishibashi N, Koide M, Uchita S, Seguchi M. When should pulmonary artery angioplasty be performed for Fontan candidates with pulmonary coarctation? Two cases of pulmonary artery angioplasty with the Blalock-Taussig shunt on pump in neonates. Jpn J Thorac Cardiovasc Surg. 2004;52(4):185-8. [MedLine]
7. Shinkawa T, Yamagishi M, Shuntoh K, Miyazaki T, Hisaoka T, Yaku H. Pulmonary arterial reconstruction for pulmonary coarctation in early infancy. Ann Thorac Surg. 2007:83(1):188-92. [MedLine]
8. De Troyer A, Yernault JC, Englert M. Lung hypoplasia in congenital pulmonary valve stenosis. Circulation. 1977;56(4 Pt 1):647-51. [MedLine]
9. Kim HK, Kim WH, Kim SC, Lim C, Lee CH, Kim SJ. Surgical strategy for pulmonary coarctation in the univentricular heart. Eur J Cardiothorac Surg. 2006;29(1):100-4. [MedLine]
10. Karamlou T, Schultz JM, Silliman C, Sandquist C, You J, Shen I, et al. Using a miniaturized circuit and an asanguineous prime to reduce neutrophil-mediated organ dysfunction following infant cardiopulmonary bypass. Ann Thorac Surg. 2005;80(1):6-13. [MedLine]
11. Wernovsky G. Improving neurologic and quality-of-life outcomes in children with congenital heart disease: past, present, and future. J Thorac Cardiovasc Surg. 2008;135(2):240-2.
12. Oliveira DM, Monteiro A, Lino FJS, Binello FCL, Colafranceschi AS. Nova tática cirúrgica para correção da coarctação da artéria pulmonar sem uso de circulação extracorpórea. Rev SOCERJ. 2009;21(supl A):32.
13. Luhmer I, Ziemer G. Coarctation of the pulmonary artery in neonates. Prevalence, diagnosis, and surgical treatment. J Thorac Cardiovasc Surg. 1993;106(5):889-94. [MedLine]
14. Troise DE, Guarnieri GF, Balducci G, Tagliente MR, Arciprete PM, Schinosa LL. Anatomical model at risk of coarctation of the pulmonary artery. Ann Thorac Surg. 2008;85(4):1505. [MedLine]
Article receive on Saturday, July 4, 2009




 All scientific articles published at rbccv.org.br are licensed under a Creative Commons license
All scientific articles published at rbccv.org.br are licensed under a Creative Commons license