INTRODUCTION
JONES et al.[1] in 1983 demonstrated paradoxical emboli by the patent foramen ovale (PFO) in a few clinical cases. In the same year, HART & MILLER [2] reported that more than one third of strokes and transitory ischemic attacks (TIA) in young adults were of unknown origin, that is to say cryptogenic. Until then the possible source of the small system emboli was considered to be prolapse of the mitral valve, smoking and primary or secondary alterations of the coagulation system.
The studies in necropsy by PATTEN [3] in 1931, of 4,083 cadavers, and by HAGEN et al., in 1984, of 965 cadavers of adolescents and adults demonstrated a prevalence of PFO of 27%. It was only in 1988 that LECHAT et al. [5], with the help of contrast transesophageal echocardiography (TEE), defined the prevalence of PFO in under 55-year-old undetermined stroke and TIA patients. This proved to be very important, affecting 54% or more of the patients. Other prospective echocardiographic studies by WEBSTER et al. [6] (50%) and JEANRENAUD et al. [7] (70%) confirmed similar high results.
As it is rarely possible to prove the passage of emboli through the PFO, in these cases paradoxical embolia was the presumed mechanism. The factor that encouraged further clinical research about undetermined strokes and TIA associated to PFO was the incidence of recurrence of strokes of up to 14.4%. This situation threatened these young adults as was demonstrated at the start of the 1990s in retrospective studies by COMESS et al. [8], STONE et al. [9] (8.4%), MAS & ZUBER [10] (3.4%) and prospective studies by BOGOUSSLAVSKY [11] (3.8%). These ratifications led several authors (DI TULLIO et al. [12], CABANES et al. [13] and HOMMA et al. [14] to define to what extent the anatomical structures of the interatrial septum could be implicated in embolic events. This aspect is of fundamental importance for the stratification of recurrence risk of strokes or TIA until the present time. In relation to this, a French study group was formed by MAS & ZUBER [10], who described the incidence of recurrence of 6.7% in the first 24 months of follow-up, with an increase to as much as 9% of the patients with PFO and aneurysms of the interatrial septum (AIAS). Diagnostic criteria had been previously defined by PEARSON et al. [15]; in which the aneurysm base and the digression away from the interatrial septum axis should exceed 15 mm and 10 mm respectively.
In 2000, DE CASTRO et al. [16] concluded that the association of a right-left shunt demonstrated during rest, without the necessity of increasing pressures using the Valsalva maneuver, and the mobility of the fossa ovale membrane of greater than 6.5 mm constitutes a recurrence risk of strokes and TIA of on average, 7.6% in the first year and 12.5% in the second year of follow-up. This association represents a significant increase in the risk of future embolic cerebral events, even greater than just the deviation that appears during the Valsalva maneuver or a limited mobility of the membrane.
The results of research using contrast TEE enabled a more precise indication of the type of prophylaxis necessary in recurrent strokes and TIA.
In 1994, MATCHAR et al. [17] and MORRISON et al. [18] demonstrated that, in patients with PFO, recurrent strokes and TIA can not be prevented totally by the classical antithrombotic pharmacological treatment, which uses anticoagulants or anti-aggregates.
Also, the first attempts of closure of the PFO as was reported by BRIDGES et al. [19] using a transcutaneously implanted prosthesis, proved to be ineffective, with as many as 10% of patients presenting with new TIA events. They also suffered from complications due to the procedure, which necessitate emergency surgical operations to remove devices that migrated either to the heart chambers or to the peripheral vascular system, as was described by AGARWAL et al. [20] in 1996.
The development and sophistication of materials employed, as well as a better definition of the morphological criteria of PFO and AIAS proposed by MARSHALL & LOCK [21], and by EWERT et al. [22] respectively, have improved the placement and the choice of the most appropriate devices.
One of the greatest problems related to the transcutaneous closure of PFO still resides in the necessity of continued anticoagulation treatment due to the formation of peri-protectic thrombosis. HOEPP et al. [23] performed a study of 20 consecutive patients who utilized anticoagulants and confirmed a prevalence of 30% of cases who suffered from thrombosis.
To avoid the deleterious effects of long-term anticoagulation in young patients with PFO and undetermined strokes and TIA, and the complications due to the transcutaneously introduced devices, a protocol to select patients who would benefit from surgical prophylaxis was established.
Thus, this investigation aims to analyze the results obtained from 47 patients submitted to surgical prophylactic treatment secondary to strokes or TIA.
METHOD
Subjects
From December 1992 to October 1998, 47 patients (31 male) with PFO and strokes or TIA of unknown origin, with ages ranging from 27 to 59 years old (mean age 40 years), were selected for direct surgical closure of the PFO in the University Hospital of Lausanne and the Hospital de la Tour in Geneva, Switzerland.
Twenty-four patients presented with at least one of the following thromboembolic risk factors: smokers (11), hypercholesterolemia (11), obesity (2) and familiar (4). One patient presented with bronchial asthma treated with terbutaline sulfate (Nº 9, Table 1).
Constituted groups
After receiving written consent from all the patients informing them of the norms of the Helsinki convention, the following selection criteria were utilized for surgical treatment:
The most important criteria were PFO diagnosed during TEE (47), stroke or TIA of unknown etiology (47) and age = 59 years (47). The less important criteria considered were at least two of the following findings: aneurysm of the interatrial septum (AIAS) (17), multiple cerebral infarctions (34), multiple cerebral events (39), Valsalva maneuver preceding strokes or TIA (12).
Procedures of clinical diagnosis
In all the patients a TEE demonstrated a PFO with a right-left arterial shunt, visible by the passage of micro-bubbles from 10 mL of isotonic saline solution injected into the basilic vein, as is illustrated in Figure 2. Simultaneously, it was possible to demonstrate by means of transcranial Doppler ultrasound (TDU) their arrival in the brain, confirming the possibility of paradoxical emboli, as is demonstrated in Figure 2. In 17 patients an aneurysm of the interatrial septum (AIAS) was demonstrated and in one patient (Nº 1, Table 1) an intrapulmonary shunt, which was not located. The criteria utilized for diagnosis of AIAS included base width > 15 mm and deviation in relation to the interatrial septum axis of 10 mm. The maximum size of the PFO was 16 mm.
Stroke was defined as an acute and permanent neurological deficit, caused by an obstructive lesion of known arterial distribution evidenced by brain magnetic resonance (BMR), as demonstrated in Figure 3 or by computed axial tomography (CAT). The TIA was defined as an acute localized and transitory neurological deficit without a corresponding lesion on the BMR or CAT. The stroke or TIA is defined as cryptogenic when its etiology is unknown and without evidence of embolic source on the TDU, BMR, CAT blood coagulation tests, TEE and 24-hour electrocardiographic monitoring. This last investigation was performed to exclude intermittent atrial fibrillation-type arrhythmia, capable of causing thromboembolism from the left atrium. The data of the patients, the diagnostic procedures and the preoperative neurological and cardiological diagnosis are demonstrated in Figures 1, 2, 3 and 4 and in Table 1 respectively.
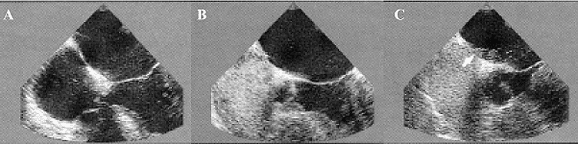
Fig. 1 - Demonstration by transesophageal echocardiography (TEE) of the right-left arterial shunt for patent foramen ovale (PFO) before (A) and after (B, C,) administration of contrast (Echovist) in patient Nº 35 of this series
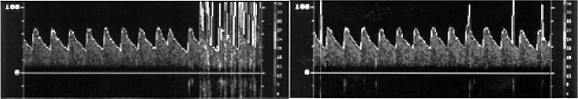
Fig. 2 - Demonstration by transcranial Doppler Ultrasonography (TDU) of the arrival of the micro-bubbles in the middle cerebral artery (arrow), after the bubbles passed by the interatrial septum during the Valsalva's maneuver in patient Nº 35 of this series
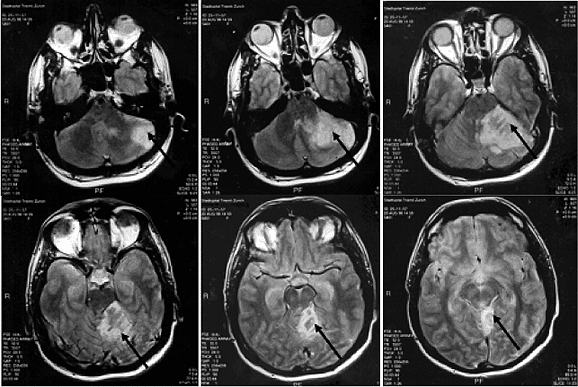
Fig. 3 - Demonstration by magnetic nuclear resonance of a ischemic infarction of the left cerebral hemisphere (arrows) after the injection of contrast in patient Nº 44 of this series
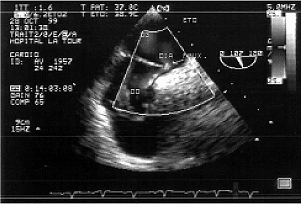
Fig. 4 - Intra-operative transesophageal echocardiography demonstrating the right-left arterial shunt (in blue) by foramen ovale, before starting cardiopulmonary bypass in patient Nº 22 of this series
Surgical procedure
All the patients were submitted to median sternotomy. For esthetic reasons, a bilateral submammary cutaneous incision was made in the youngest patient (Nº 46, Table 1). Cardiopulmonary bypass (CPB) was established after systemic heparinization, with introduction of the catheters in the ascending aorta and vena cavas. Thirty operations occurred at moderate hypothermia (32º rectum temperature) without cardioplegia, inducing slight ventricular fibrillation after aortic clamping. The PFO was identified after right atriotomy. In 46 patients atrioseptorraphia was performed, posterior to Todaro's tendon, with continuous suturing using 4-0 polypropylene thread. Only in one case (Nº 7, Table 1) separate sutures were used. In patients with AIAS, the fold was achieved with the same suturing. The atriotomy was closed with a 4-0 polypropylene thread. After removal of air from the heart chambers, the aorta was de-clamped. The CPB was terminated when the rectal temperature reached 35º C. Before administering protamine, a routine check of the closure of the interatrial septum was performed using an intraoperative TEE (Figures 4 and 5).
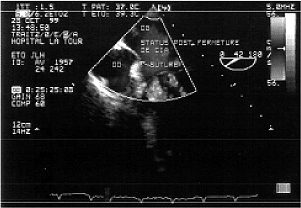
Fig. 5 - Intra-operative transesophageal echocardiography after removing the cardiopulmonary bypass demonstrating the absence of residual shunts in the interatrial septum of patient Nº 22 of this series confirming the success of atrioseptorrafia
Postoperative follow up
Before release from hospital, a thoracic radiogram, ECG and TEE were obtained. In an average period of 36 months (from 7 to 100 months), all the patients underwent a clinical re-evaluation with transcranial Doppler echogram combined with contrast TEE and the Valsalva maneuver. A thoracic radiogram and ECG were repeated, as well as BMR to investigate possible silent infarctions, which could have occurred during the follow-up period.
RESULTS
The mean time from the neurological diagnosis (Table 1) to the surgical closure of the PFO was 58.1 days (from 7 to 210 days). The patients were operated on by six different surgeons and the operative time was from 100 to 165 minutes (mean 145 minutes). The mean times of aortic clamping and of CPB were 13 minutes (from 6 to 24 minutes) and 28 minutes (from 22 to 42 minutes) respectively, as is shown in Table 2. The sinusal rhythm reappeared spontaneously in 42 patients and after internal electric shock of 10 joules in five. Some hours after the operation, four patients developed transitory arrhythmias (three nodal rhythms and one ventricular tachycardia, which were immediately reverted to sinusal rhythm by the injection of lidocaine in bolus).
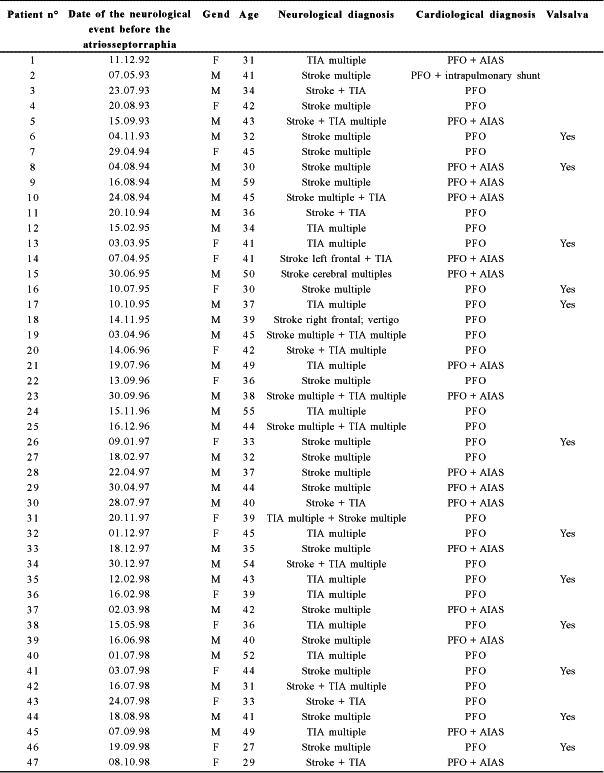
Table 2. Operative times
CPB = cardiopulmonary bypass; min. = minutes
No embolic events, atrial fibrillation or hemodynamic instability were seen. Twelve patients received dopamine at 5 ìg/ kg/min during the first 12-24 hours. The patients stayed in the intensive care unit on average for 19.6 hours (from 8 to 24 hours). Only one patient required 600 mL of autologous blood transfusion (Nº 17, Table 1). Thoracic drainage varied from 265 to 670 mL (mean 410 mL). The thoracic drains were removed within 30 to 48 hours (mean of 42 hours). Recovery of all the patients occurred without complications, except for the youngest patient (Nº 46, Table 1) who needed to be readmitted to an intensive care unit of another hospital, three days after release, owing to a significant drop of methaemoglobinaemia (6 g/L).
Pleural stokes, residual intracardiac veno-arterial shunts, atrioventricular blocks, strokes or TIA were not observed during the mean hospitalization period of 8.5 days (from 8 to 10 days). One patient (Nº 11, Table 1), who had previously presented with psychotic episodes, presented with behavioral disturbances two months after the operation, without evidence of simultaneous embolic events. At 36 months, no cases of strokes or TIA or extracranial embolic events were evidenced and the BMR did not demonstrate new lesions. No patient presented thrombosis in the right atrium or at the edges of the persistent foramen ovale.
The only patient (Nº 7, Table 1) in whom a residual right-left shunt was found with micro-bubbles in the Valsalva test, atrioseptorraphia was performed with separate sutures. The pre-operative shunt had a large number of micro-bubbles. Although a residual interatrial shunt was excluded with the help of contrast TEE, in the patient with an intrapulmonary shunt (Nº 2, Table 1), transcranial doppler echography evidenced a small late passage of contrast (> 20 micro-bubbles), compatible with this type of extracardiac shunt.
In 17 patients with corrections of the AIAS, residual deviation of the interatrial septum was evidenced.
The thoracic radiogram and the ECG did not demonstrate alterations in this series.
COMMENTS
The prevalence of PFO was described by LECHAT et al. [5] at 40% of young adults with strokes or TIA of unknown etiology, but DE BELDER et al. [24] demonstrated that the PFO constitutes a thromboembolic risk factor in all age groups. For this, it was decided that patients with up to 59 years of age were included in this study. Additionally, the selection criteria demanded by our protocol allowed the exclusion of patients with other potential causes of strokes or TIA. Patients who presented with multiple cerebral events, history of Valsalva maneuver or AIAS were also selected for operations, as these factors might be associated with high risk of reoccurrence, when compared with the risk in other patients with strokes or TIA and PFO (CABINES et al. [13]; ZUBER & MAS [25]).
As already pointed out, currently the most common treatment to reduce the frequency of recurrent paradoxical embolia is anticoagulation, but the prognosis of patients with strokes or TIA, submitted to long-term anticoagulation, is harmed due to hemorrhagic complications, specifically cerebral complications (BOGOUSSLAVSKY & REGLI [26]; KOSTANTINIDES & JUST [27]). For these reasons, anticoagulation is indicated in two specific clinical situations: whilst waiting for the surgical closure of the PFO and to treat venous thrombosis. Prophylaxis of recurrent strokes of unknown origin only with the use of platelet anti-aggregates seems to be inappropriate as paradoxical embolism also depends on venous thrombosis, which principally demands anticoagulation and not anti-aggregates. Transcutaneous introduced devices constitute a therapeutic alternative, but must frequently be associated to anticoagulation, especially when there is recurrence of strokes or TIA (BRIDGES et al. [19]; RAO et al. [28]) and also because of the presence of a significant number of cases of peri-protectic thrombosis (HOEPP et al. [23]). Despite of the possibility of recovering some devices using available catheters, WINDECKER & MEIER [29] in 1999 reported innumerous disadvantages of these systems. These disadvantages include right atrial disc obstructed with the "Amplatzer PFO occluder"; difficulty of repositioning of the "CardioSEAL septal occluder", the "PFO STAR", and the "DAS occluder"; Septal necrosis with the "ASDOS device" and embolic events with the "Sideris Buttoned occluder". Thus, the interventionalist treatment can still be considered as experimental, which should be further studied in centers trained in this type of procedure, with the selection of patients respecting defined protocols.
The current tendency, according to DEVUYST & BOGOUSSLAVSKY [30] in 2000 is to introduce the devices when, apart from the principal criteria, two less important criteria are also evidenced; on the other hand, surgical treatment should be indicated when three less-important criteria are present. Although these devices have still not been approved for clinical application by the Food and Drug Administration or the European Community, a comparative analysis of interventionalist costs and respective surgical treatment of interatrial communications, performed by BAKER et al. [31] in 2002 described the current tendency to favor prostheses as opposed to surgery for economic reasons. The therapeutic abstention in cases of paradoxical embolism through the PFO would be the worst option, as the risk of recurrence exceeds 0.8% per year, according to the stratification of risk proposed by NENDAZ et al. [32].
Table 3 presents the comparative results of the different types of treatment for strokes and TIA of unknown origin, showing the advantages of surgical treatment. This treatment has the potential of eradicating the necessity of intracardiac prostheses, anticoagulation with its problems related to the compliance of patients and also the potential of eliminating recurrent paradoxical embolism. In this series, the only residual shunt was probably the result of the technique of suturing with separate stitches, which suggests that continuous suturing is better indicated. After this experience, it was decided to close the PFO systematically with double continuous suturing.
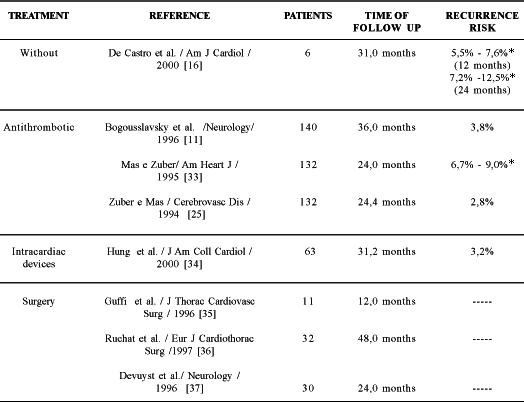
Table 3. Recurrence risk of stroke or transitory ischemic attacks in patients with patent foramen ovale and proved cerebral ischemia according to the type of treatment.
* with aneurysm of the interatrial septum
Gaseous embolism constitutes the greatest danger in patients submitted to operations for the correction of interatrial septum defects (KIRKLIN & BARRATT-BOYES [38] in 1993). This might be avoided minimizing the intra-operative suction of atrial blood, suction of air at the aortic root before electric cardioversion or maintaining the filling of the left atrium by a continuous flow of blood from the pulmonary veins, after induction of fine ventricular fibrillation. This last technique dispenses with the use of cardioplegia, allowing rapid re-establishment of cardiac output, and was also successfully utilized in four patients by TEVEARAI et al. [39].
The times of surgery, perfusion and aortic clamping related to this little invasive approach exceed the times employed in the classic median sternotomy. It is an interesting technique, but it should not be considered as a routine procedure and substitute the usual technique, which gives us well-defined safety margins. Atrioseptorraphia can also be performed using a little evasive robotic technique assisted by video whose promising results were recently published by TORRACCA et al. [40].
Although in this series, no deaths occurred and the morbidity was minimal, randomized prospective studies need to be made to assess the long-tern efficacy of the surgical approach when compared with pharmacological and interventionalist treatments. The hospital stay was long in relation to the standard treatment. Nevertheless this allowed a more detailed follow up, especially in relation to the cardiac and cerebral ultrasound control searching for short-term residual shunts. In fact the patients were studied with TEE between the seventh and tenth post-operative day, when it is supposed that the process of endothelium regeneration is defined. Recurrence of strokes or TIA after atriosseptorraphia for PFO might not be related to paradoxical embolism, but with another etiology of strokes and TIA, which might be identified later (DEARANI et al. [41], 1999), when no interatrial shunt or residual AIAS exists. We are enthusiastic about the results presented in this work, and 25 additional patients, selected according to this protocol, were also operated on with success, and they were also released from hospital in good clinical conditions without interatrial veno-arterial shunts.
Surgical prophylaxis secondary to strokes or TIA of unknown origin, in the 47 patients selected with PFO, demonstrated to be safe and no patient presented with strokes or TIA during the study period.
CONCLUSIONS
Surgical prophylaxis secondary to strokes or transitory ischemic attacks in patients with persistent foramen ovale demonstrated to be efficient without any deaths, functional sequels nor relapses occurring during the investigative period.
ACKNOWLEDGEMENTS
This work was possible owing to the encouragement and collaboration of helpful colleagues in several sectors of cardiology (Dr. Jeanrenaud Xavier, of the University Hospital of Lausanne and Dr. Schopfer Patric, of the Hospital de la Tour of Geneva), of neurology (Prof. Dr. Bogousslavsky Jean, of the University Hospital of Lausanne) and of cardiac surgery (Dr. Velebit Vladimir, of the Hospital de la Tour of Geneva, and Prof. Dr. Sadeghi Houssein, of the University Hospital of Lausanne). To Prof. Dr. Otoni Moreira Gomes, for his friendship and giving value to this work with untiring help.
BIBLIOGRAPHIC REFERENCES
1 Jones Jr. HR, Caplan LR, Come PC, Swinton Jr. NW, Breslin DJ. Cerebral emboli of paradoxical origin. Ann Neurol 1983; 13: 314-9.
[ Medline ]
2 Hart RG, Miller VT. Cerebral infarction in young adults: a practical approach. Stroke 1983; 14:110-4.
[ Medline ]
3 Patten BM. The closure of the foramen ovale. Am J Anat 1931; 48:19-44.
4 Hagen PT, Scholz DG, Edwards WD. Incidence and size of patent foramen ovale during the first 10 decades of life: an autopsy study of 965 normal hearts. Mayo Clin Proc 1984; 59:17-20.
[ Medline ]
5 Lechat P, Mas JL, Lascault G, Loron P, Theard M, Klimczac M et al. Prevalence of patent foramen ovale in patients with stroke. N Engl J Med 1988; 318:1148-52.
[ Medline ]
6 Webster MW, Chancellor AM, Smith HJ, Swift DL, Sharpe DN, Bass NM et al. Patent foramen ovale in young stroke patients. Lancet 1988; 2:11-2.
7 Jeanrenaud X, Bogousslavsky J, Payot M, Regli F, Kappenberger L. Patent foramen ovale and cerebral infarct in young patients. Schweiz Med Wochenschr 1990; 120:823-9.
[ Medline ]
8 Comess KA, DeRook FA, Beach KW, Lytle NJ, Golby AJ, Albers GW. Transesophageal echocardiography and carotid ultrasound in patients with cerebral ischemia: prevalence of findings and recurrent stroke risk. J Am Coll Cardiol 1994;23:1598-603.
[ Medline ]
9 Stone DA, Godard J, Corretti MC, Kittner SJ, Sample C, Price TR et al. Patent foramen ovale: association between the degree of shunt by contrast transesophageal echocardiography and the risk of future ischemic neurologic events. Am Heart J 1996;131:158-61.
[ Medline ]
10 Mas JL, Zuber M. Recurrent cerebrovascular events in patients with patent foramen ovale, atrial septal aneurysm, or both and cryptogenic stroke or transient ischemic attack. French Study Group on Patent Foramen Ovale and Atrial Septal Aneurysm. Am Heart J 1995; 130:1083-8.
[ Medline ]
11. Bogousslavsky J, Garazi S, Jeanrenaud X, Aebischer N, Van Melle G. Stroke recurrence in patients with patent foramen ovale: the Lausanne Study. Lausanne Stroke with Paradoxal Embolism Study Group. Neurology 1996; 46:1301-5.
12. Di Tullio M, Sacco RL, Venketasubramanian N, Sherman D, Mohr JP, Homma S. Comparison of diagnostic techniques for the detection of a patent foramen ovale in stroke patients. Stroke 1993; 24:1020-4.
[ Medline ]
13. Cabanes L, Mas JL, Cohen A, Amarenco P, Cabanes PA, Oubary P et al. Atrial septal aneurysm and patent foramen ovale as risk factors for cryptogenic stroke in patients less than 55 years of age. A study using transesophageal echocardiography. Stroke 1993; 24:1865-73.
14. Homma S, Di Tullio MR, Sacco RL, Mihalatos D, Li Mandri G, Mohr JP. Characteristics of patent foramen ovale associated with cryptogenic stroke. A biplane transesophageal echocardiographic study. Stroke 1994; 25:582-6.
15. Pearson AC, Nagelhout D, Castello R, Gomez CR, Labovitz AJ. Atrial septal aneurysm and stroke: a transesophageal echocardiographic study. J Am Coll Cardiol 1991; 18:1223-9.
[ Medline ]
16. De Castro S, Cartoni D, Fiorelli M, Rasura M, Beni S, Urani C et al. Patent foramen ovale and its embolic implications. Am J Cardiol. 2000; 86:51G-52G.
17. Matchar DB, McCrory DC, Barnett HJ, Feussner JR. Medical treatment for stroke prevention. Ann Intern Med 1994;121:41-53.
[ Medline ]
18. Morrison BJ, Landzberg MJ, Newburger JW, Jenkins KJ, Faherty C, Lock JE. Infrequent occurrence of recurrent paradoxical embolism at intermediate follow-up after trans-catheter closure of patent foramen ovale. Circulation 1994; 90(Suppl):I237.
19. Bridges ND, Hellenbrand W, Latson L, Filiano J, Newburger JW, Lock JE. Transcatheter closure of patent foramen ovale after presumed paradoxical embolism. Circulation 1992; 86:1902-8.
[ Medline ]
20. Agarwal SK, Ghosh PK, Mittal PK. Failure of devices used for closure of atrial septal defects: mechanisms and management. J Thorac Cardiovasc Surg 1996; 112:21-6.
[ Medline ]
21. Marshall AC, Lock JE. Structural and compliant anatomy of the patent foramen ovale in patients undergoing transcatheter closure. Am Heart J 2000; 140(2):303-7.
[ Medline ]
22. Ewert P, Berger F, Vogel M, Dahnert I, Alexi-Meshkishvili V, Lange PE. Morphology of perforated atrial septal aneurysm suitable for closure by transcatheter device placement Heart 2000; 84:327-31.
23. Hoepp HW, Deutsch HJ, La Rosee K, Schnabel P, Terheggen G, Schneider CA et al. Transcatheter closure of atrial-septal defects and patent foramen ovale in adults: optimal anatomic adaptation of occlusion device.Am Heart J 1999; 138(5 Pt 1):941-9.
[ Medline ]
24. De Belder MA, Tourikis L, Leech G, Camm AJ. Risk of patent foramen ovale for thromboembolic events in all age groups. Am J Cardiol 1992; 69:1316-20.
[ Medline ]
25. Zuber M, Mas JL: Risk of recurrent stroke in patients with atrial septal aneurysm or patent foramen ovale: a French multicentric study. Cerebrovasc Dis 1994; 4:247.
26. Bogousslavsky J, Regli F. Anticoagulant-induced intracerebral bleeding in brain ischemia: evaluation in 200 patients with TIAs, emboli from the heart, and progressing stroke. Acta Neurol Scand 1985; 71:464-71.
[ Medline ]
27. Konstantinides S, Just H. Patent foramen ovale: conservative or surgical therapy? Z Kardiol 2000; 89:63-71.
[ Medline ]
28. Rao PS, Wilson AD, Chopra PS. Transcatheter closure of atrial septal defect by "buttoned" devices. Am J Cardiol 1992; 69:1056-61.
[ Medline ]
29. Windecker S, Meier B. Percutaneous patent foramen ovale (PFO) closure: it can be done but should it? Catheter Cardiovasc Interv 1999; 47:377-80.
30. Devuyst G, Bogousslavsky J. Indications à la fermeture du foramen ovale perméable. Med Hyg 2000; 58:1306-9.
31. Baker SS, O'Laughlin MP, Jollis JG, Harrison JK, Sanders SP, Li JS. Cost implications of closure of atrial septal defect. Catheter Cardiovasc Interv 2002; 55:83-7.
32. Nendaz MR, Sarasin FP, Junod AF, Bogousslavsky J. Preventing stroke recurrence in patients with patent foramen ovale: antithrombotic therapy, foramen closure, or therapeutic abstention? A decision analytic perspective. Am Heart J 1998; 135:532-41.
[ Medline ]
33. Mas JL, Zuber M. Recurrent cerebrovascular events in patients with patent foramen ovale, atrial septal aneurysm, or both and cryptogenic stroke or transient ischemic attack. French Study Group on Patent Foramen Ovale and Atrial Septal Aneurysm. Am Heart J 1995; 130:1083-8.
[ Medline ]
34. Hung J, Landzberg MJ, Jenkins KJ, King ME, Lock JE, Palacios IF et al. Closure of patent foramen ovale for paradoxical emboli: intermediate-term risk of recurrent neurological events following transcatheter device placement.J Am Coll Cardiol 2000; 35:1311-6.
[ Medline ]
35. Guffi M, Bogousslavsky J, Jeanrenaud X, Devuyst G, Sadeghi H. Surgical prophylaxis of recurrent stroke in patients with patent foramen ovale: a pilot study. J Thorac Cardiovasc Surg 1996; 112:260-3.
[ Medline ]
36. Ruchat P, Bogousslavsky J, Hurni M, Fischer AP, Jeanrenaud X, von Segesser LK. Systematic surgical closure of patent foramen ovale in selected patients with cerebrovascular events due to paradoxical embolism. Early results of a preliminary study. Eur J Cardiothorac Surg 1997; 11:824-7.
[ Medline ]
37. Devuyst G, Bogousslavsky J, Ruchat P, Jeanrenaud X, Despland PA, Regli F et al. Prognosis after stroke followed by surgical closure of patent foramen ovale: a prospective follow-up study with brain MRI and simultaneous transesophageal and transcranial Doppler ultrasound. Neurology 1996; 47:1162-6.
[ Medline ]
38. Kirklin JW, Barratt-Boyes BG. Atrial septal defect and partial anomalous venous connection. In: Kirklin JW, Barratt-Boyes BG, eds. Cardiac surgery. 2nd ed. New York: Churchill Livingstone, 1993. p. 634-5.
39. Tevaearai HT, Mueller XM, Ruchat P, Hurni M, Stumpe F, Fischer A et al. "Minimally invasive" surgical closure of the patent foramen ovale. Swiss Surg 1998; Suppl 2:14-7.
40. Torracca L, Ismeno G, Alfieri O. Totally endoscopic computer-enhanced atrial septal defect closure in six patients.Ann Thorac Surg 2001;72:1354-7.
[ Medline ]
41. Dearani JA, Ugurlu BS, Danielson GK, Daly RC, McGregor CG, Mullany CJ, et al. Surgical patent foramen ovale closure for prevention of paradoxical embolism-related cerebrovascular ischemic events. Circulation 1999; 100 (19 Suppl):II171-5.








 All scientific articles published at rbccv.org.br are licensed under a Creative Commons license
All scientific articles published at rbccv.org.br are licensed under a Creative Commons license