Objective: Patients with functional single ventricle show bad survey, most of which result in cardiac failure even in the case that they are operated. The right heart bypass operations except for right atrium and right ventricle connections lack pulsatile pulmonary ventricle system besides the volume lack coming to the common ventricle. To solve the problem, we tried to create a pulmonary ventricle which produces pulsatile pressure experimentally.
Methods: We tried to form a right ventricle system which produces pulsatile pressure. The experimental model was carried on six sheep. The pressures of the created pulmonary ventricle, pulmonary artery and the left ventricle were measured after leaving the cardiopulmonary bypass.
Results: The mean of the systolic and diastolic pulmonary artery pressures were 15,6 ± 2.0 mmHg and 4.5 ± 1.5 mmHg. The mean of the left ventricular systolic pressure was mean 76.6 ± 4.4 mmHg.
Conclusion: A ventricle producing pulsatile pressure is necessary for regulating the pulmonary artery flow with high central venous pressure and low non-pulsatile pulmonary pressure in the anomalies with functional single ventricles.
Objetivo: Pacientes com ventrículo único funcional têm prognóstico ruim, que resulta em insuficiência cardíaca, mesmo após tratamento cirúrgico. As operações derivação cavo-pulmonar (exceto pelas conexões do átrio direito ao ventrículo direito) não apresentam sistema ventricular pulmonar com pressão pulsátil, além do fluxo reduzido ao ventrículo único. Para resolver o problema, tentamos criar um ventrículo pulmonar que produza pressão pulsátil experimentalmente.
Método: Tentamos criar um sistema ventricular direito que produzisse pressão pulsátil. O modelo experimental foi realizado em seis ovelhas. As pressões do ventrículo pulmonar criado, da artéria pulmonar e do ventrículo esquerdo foram medidas após a saída da circulação extracorpórea.
Resultados: A média das pressões arteriais pulmonares sistólica e diastólica foi 15,6 ± 2,0 mmHg e 4,5 ± 1,5 mmHg. A média da pressão sistólica ventricular esquerda foi 76,6 ± 4,4 mmHg.
Conclusão: Um ventrículo que produza pressão pulsátil é necessário para a regulação do fluxo da artéria pulmonar, com pressão venosa central e pressão pulmonar não pulsátil nas anomalias como ventrículos únicos funcionais.
INTRODUCTION
The term single ventricle refers to congenital cardiac anomalies in which there is only one functional single ventricular chamber giving rise to the great vessels. It may occur due to a failure of the ventricular septation or hypoplasia of one of the ventricles. Some other congenital heart defects may also share the characteristics of a functionally single ventricle for example tricuspid atresia. Surgical therapy consists of palliative procedures such as pulmonary artery banding and systemic arterial to pulmonary artery shunting and definitive repair procedures such as right heart bypass and ventricular septation procedures. The septation procedure is rarely performed [1].
Patients with functionally single ventricles have bad prognosis usually resulting in heart failure even in operated cases. Patients to whom right heart bypass operations had been applied have a 73% 15-year survival under optimum conditions (except for right atrium to right ventricular connections) [2]. Right heart bypass operations, except for right atrium and right ventricle connections, lack a pulsatile pulmonary ventricle system and lack volume returning to the common ventricle. This results in a high central venous pressure. To solve this problem, we tried to experimentally create a pulmonary ventricle which produces pulsatile pressure.
METHODS
In order to create a pulsatile pulmonary ventricle system, we designed a model. In this model, the right heart would be bypassed and there would be an artificial pulmonary ventricle system which would take the place of the right ventricle (Figure 1). This model was demonstrated in six sheep hearts under open heart surgery conditions. The sheep weighed 40 to 45 kg and their ages were from 1.5 to 2 years old. Sheep are preferred for the experimental model due to the fact that they are common in our country and sheep are an appropriate animal model for cardiac research [3-5]. The study was approved by the local Ethics Committee. The experiment was performed according to the Ethics Committee on Animal Research, Ege University Medical School, Ýzmir, Turkey. The study was funded by the authors. The animals were sacrificed after completion of the hemodynamic measurements.
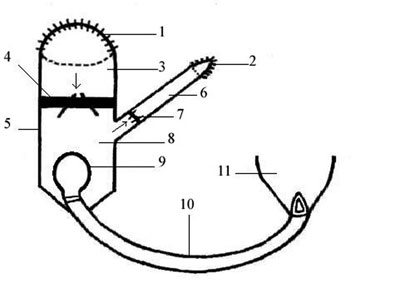
Fig. 1 - The experimental model for a pulmonary ventricle which produces pulsatile pressure. A 29-mm mechanical heart valve was placed into a 30-mm, 15-cm long Dacron graft with 2-0 running silk sutures in a way that it would open to two thirds of the graft diameter. One third of the graft was anastomosed to the right atrium of the patient with 3-0 polypropylene sutures (Figures 1 and 3).
The pulmonary artery connection of the system mentioned above was provided by an 8-mm Dacron graft. A 19-mm mechanical heart valve was placed into the graft.
1) The Right atrial anastomosis on the 30-mm Dacron graft, 2) The pulmonary arterial anastomosis of the 8-mm Dacron graft, 3) Right atrial side of the system, 4) 29-mm mechanical heart valve, 5) 30-mm Dacron graft, 6) 8-mm Dacron graft, 7) 19-mm mechanical heart valve, 8) Artificial pulmonary ventricle, 9) Elastic (balloon-like) material which was fixed onto the rear end of the no 28 cannula, 10) no 28 cannula, 11) Left ventricular apex. The arrows indicate the flow direction.
Six sheep underwent heart surgery following general anesthesia initiated with xylasine. The sheep were submitted to median sternotomy. After cannulation of the aorta, bicaval venous cannulations were performed. The cavas were controlled by the application of snares. The surgery was performed on the beating heart using cardiopulmonary bypass. The aorta of the sheep was not clamped.
An elastic (balloon-like) material, whose diameter can change with pressure, was fixed onto the end of a no. 28 venous cannula. When this cannula was inserted in to the left ventricular apex, it was seen that the balloon inflated on each systole (Figures 1 and 2).
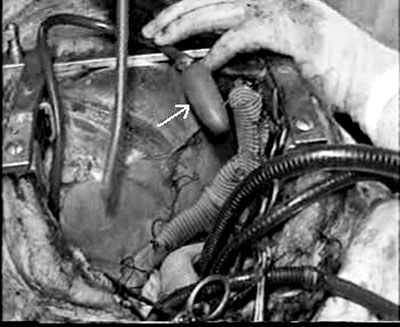
Fig. 2 - An elastic (balloon-like) material, whose diameter can change with pressure, was fixed onto the end of the no. 28 venous cannula. When this cannula was inserted in the left ventricle apex, the balloon was seen to inflate on each systole. The arrow indicates the balloon which was connected to the left ventricular apex using a no 28 cannula.
In order to create a pulmonary ventricle and a right atrium, a mechanical 29-mm mechanical heart valve was placed into a 30-mm, 15-cm long Dacron graft with 2-0 running silk sutures in a way that it would open to two thirds of the graft diameter. One third of the graft was anastomosed to the right atrium of the patient with 3-0 polypropylene sutures (Figures 1 and 3).
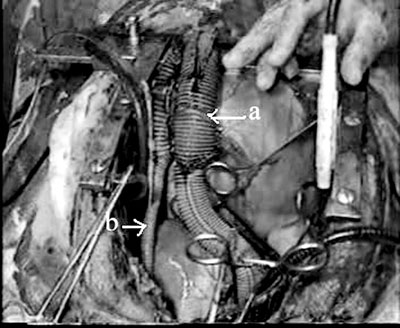
Fig. 3 - Picture of the artificial pulmonary ventricle (a). The pulmonary artery connection of the artificial pulmonary ventricle via an 8-mm Dacron graft (b).
The pulmonary artery connection of the system mentioned above was provided by an 8-mm Dacron graft. A 19-mm mechanical heart valve was placed into the graft. The valve insertion point of the graft was enlarged using a Dacron patch. One end of the graft was anastomosed to the artificial pulmonary ventricle and the other end to the pulmonary artery of the sheep in a way that the valve permitted flow to the pulmonary artery.
The elastic part of the no. 28 venous cannula which inflated on systole and deflated on diastole was inserted to a position 2/3 along the 30-mm Dacron graft and the graft was secured over the venous cannula using a silk suture. Thus, a ventricle producing pulsatile flow was obtained (Figures 1 and 3).
After completion of the operation, the cardiopulmonary bypass was removed. Pressure monitoring lines were placed into the left ventricle and the pulmonary artery. Then the pulmonary artery was clamped and the right heart bypass was initiated. Simultaneous left ventricle and pulmonary artery pressures were noted.
RESULTS
The mean systolic and diastolic pulmonary artery pressures were 15.6 ± 2.0 mmHg and 4.5 ± 1.5 mmHg respectively when the mean systolic LV pressure was 76.6 ± 4.4 mmHg. The pressures measured in the pulmonary arteries of the sheep were pulsatile.
DISCUSSION
The pathology of mortality after right heart bypass operations (except for RA-RV connections) seems to be the non-pulsatile pulmonary flow and lack of volume in the ventricle which in fact causes chronic volume overload. Pulsatile pulmonary blood flow plays an important role in regulating shear-stress-mediated release of endothelium-derived nitric oxide and endothelin-1. It is also important in reducing pulmonary vascular resistance by passive capillary recruitment. A long-term non-pulsatile flow was show to lead to endothelial dysfunction. This may be involved in distention and vascular remodeling due to the increase in pulmonary vascular resistance. Non-pulsatile flow can lead to increased patency of the arteriovenous shunt, which might be, at least in part, involved in the development of pulmonary arteriovenous fistula and cause limitations after the Fontan operation [6]. A pulsatile bidirectional cavopulmonary shunt was reported to be useful for high-risk Fontan patients not only in the staged Fontan operation, but also as a definitive palliation [7].
Therefore a pulmonary ventricle producing pulsatile pressure is necessary to regulate the pulmonary arterial flow with a high central venous pressure and low non-pulsatile pulmonary pressure. So the ventricle system mentioned above will be of great importance to solve the problems of lack of volume and non-pulsatile pulmonary flow. There will probably be many difficulties to apply this system to humans. However, this experimental model showed that a pulmonary ventricle producing a pulsatile pulmonary flow is possible in a single functional ventricle.
CONCLUSION
In conclusion, the creation of a pulmonary ventricle which produces a pulsatile pulmonary flow will solve the problems encountered by a non-pulsatile pulmonary flow although the application of this experimental model may bring other problems which are not discussed in this study. The human-applicable form of this method is still under investigation.
REFERENCES
1. Fontana GP, Permut LC, Laks H. Surgical management of complex functional single ventricle. In: Baue E, Geha AS, Hammond GL, Laks H, Naunheim KS, eds. Glenn's thoracic and cardiovascular surgery. 6th ed. Stanford:Appleton & Lange;1996. p.1193-201.
2. Kouchhoukos NT, Blackstone EH, Doty BD, Hanley FL, Karp RB. Tricuspid atresia and management of single-ventricle physiology. In: Kirklin JW, Barratt-Boyes BG eds. Cardiac surgery. 3rd ed. Philadelphia:Churchill Livingstone;2003. p.1113-76.
3. Ali ML, Kumar SP, Bjornstad K, Duran CM. The sheep as an animal model for heart valve research. Cardiovasc Surg. 1996;4(4):543-9.
4. Lansac E, Lim HS, Shomura Y, Lim KH, Goetz W, Rice NT et al. Aortic and pulmonary root: are their dynamics similar? Eur J Cardiothorac Surg. 2002;21(2):268-75.
5. Lansac E, Lim HS, Shomura Y, Lim KH, Rice NT, Goetz W et al. A four-dimensional study of the aortic root dynamics. Eur J Cardiothorac Surg. 2002;22(4):497-503.
6. Yin Z, Wang Z, Zhu H, Zhang R, Wang H, Li X. Experimental study of effect of Fontan circuit on pulmonary microcirculation. Asian Cardiovasc Thorac Ann. 2006;14(3):183-8.
7. Miyaji K, Shimada M, Sekiguchi A, Ishizawa A, Isoda T. Usefulness of pulsatile bidirectional cavopulmonary shunt in high-risk Fontan patients. Ann Thorac Surg. 1996;61(3):845-50.



 All scientific articles published at rbccv.org.br are licensed under a Creative Commons license
All scientific articles published at rbccv.org.br are licensed under a Creative Commons license