Jarbas Jakson DinkhuysenI; Carlos ContrerasII; Reginaldo CipulloIII; Marco Aurélio FingerIV; João RossiIII; Ricardo ManriqueV; Hélio M. MagalhãesVI; Paulo ChaccurII
DOI: 10.5935/1678-9741.20110055
ABSTRACT
BACKGROUND AND OBJECTIVE: We attempt to reduce the ischemic time during implantation of the donor heart in the bicaval bipulmonary orthotopic position using normothermic beating heart and thus, facilitate the transplanted heart adaptation to the recipient. This study presents a small experience about a new strategy of myocardial protection during heart transplant.
METHODS: In cardiopulmonary bypass, the aorta anastomosis was done first, allowing the coronary arteries to receive blood flow and the recovering of the beats. The rest of the anastomosis is performed on a beating heart in sinus rhythm. The pulmonary anastomosis is the last to be done. This methodology was applied in 10 subjects: eight males, age 16-69 (mean 32.7 years), SPAo 90-100 mmHg (mean 96 mmHg), SPAP 25-65 mmHg (mean 46.1 mmHg), PVR 0.9 to 5.0 Wood (mean 3.17 Wood), GTP 4-13 mmHg (mean 7.9 mmHg), and eight male donors, age 15-48 years (mean 27.7 years), weight 65-114 kg (mean 83.1 kg). Causes of brain coma: encephalic trauma in five hemorrhagic stroke in four, and brain tumor in one.
RESULTS: The ischemic time ranged from 58-90 minutes (mean 67.6 minutes) and 8 donors were in hospitals of Sao Paulo and two in distant cities. All grafts assumed the cardiac output requiring low-dose inotropic therapy and maintained these conditions in the postoperative period. There were no deaths and all were discharged. The late evolution goes from 20 days to 10 months with one death occurred after 4 months due to sepsis.
CONCLUSION: This method, besides reducing the ischemic time of the procedure, allows the donated organ to regain and maintain their beats without pre or after load during implantation entailing the physiological recovery of the graft.
RESUMO
INTRODUÇÃO E OBJETIVO: Trata-se do implante em posição ortotópica bicaval bipulmonar do coração doado batendo em normotermia. Este estudo busca diminuir o tempo isquêmico e criar condições de ir se adaptando ao organismo hospedeiro.
MÉTODOS: Já em CEC, a primeira anastomose a ser feita é a da aorta, reperfundindo as artérias coronárias e recuperando os batimentos. As restantes são realizadas com o coração batendo em ritmo sinusal, sendo a da artéria pulmonar a última. Esta metodologia foi aplicada em 10 pacientes receptores, sendo oito do sexo masculino, com idades entre 16 e 69 (média de 32,7 anos), PSAo 90-100 (média de 96 mmHg), PSAP 25-65 (média de 46,1 mmHg), RVP 0,9-5,0 (média de 3,17 Wood), GTP 4-13 (média de 7,9 mmHg). Entre os 10 doadores, sete eram do sexo masculino, com idade entre 15 e 48 (média 27,7 anos), peso entre 65 e 114 kg (média de 83,1 kg). As causas do coma encefálico foram: TCE (cinco), AVCH (quarto) e tumor cerebral (um).
RESULTADOS: O tempo isquêmico variou de 58 a 90 minutos (média 67,6 minutos), sendo que oito doadores estavam em hospitais da região metropolitana de São Paulo e dois em cidades distantes. Todos os enxertos, após completadas as anastomoses, retomaram o fluxo e o débito, mantendo bons parâmetros, com baixa dosagem de inotrópico e mantiveram estas condições no pós-operatório imediato. Não ocorreram óbitos e todos os pacientes obtiveram alta hospitalar. A evolução tardia variou de 20 dias a 10 meses, tendo ocorrido um óbito ao 4º mês pós-transplante, por sepse.
CONCLUSÕES: Esta metodologia, além de reduzir o tempo isquêmico, permite ao órgão doado recuperar e manter seus batimentos sem pré nem pós-carga durante o implante, o que enseja proporcionar recuperação fisiológica, ultraestrutural, imunológica, inflamatória e mecânica do enxerto, com resultados consistentes precoces e tardios.
INTRODUCTION
The effective myocardial protection is a commitment that lies between two opposite strategies, reducing the metabolic needs by cardiac arrest at low temperatures, and increasing the myocardial temperature during the arrest, to increase the chances of immediate recovery of cardiac function. The crystalloid cardioplegia at 4ºC obviously reduces the metabolic needs; however, it is not enough to provide adequate protection [1] and warm reperfusion that provides immediate recovery of cardiac function which reverses protection induced by cold crystalloid cardioplegia [2].
Despite advances in heart transplantation, the lack of donors is still a reality, and, whenever possible, the ideal situation is to capture the graft preferably without long ischemic times. The various protocols of donor organ preservation using hypothermic arrest induced by various crystalloid cardioplegic solutions, however, it is the suboptimal state associated with long ischemic times.
The ischemic damage contributes to the risk of primary graft dysfunction and limits from 4 to 6 hours the safe period between stopping the beating heart donor and recovering the beating already implanted in the receiver. Several studies have been developed to improve results and, in 1991, Lichtenstein et al. [3] introduced the normothermia with a beating heart and other authors [4,5] followed the technique. On the other hand, blood cardioplegia has been suggested as the most appropriate method to preserve the donor heart [6] and continuous infusion with a beating heart extends the time and helps the recovery of the contractile function [7,8].
The hypothesis on this study is that, the earlier coronary reperfusion is performed at normothermia with the recovery of heart rate without the presence of pre-or post-load during the heart transplant, the better the results will be.
METHODS
In the Medical Section of Transplant at IDPC/SP, 288 cardiac transplants were performed in the period from November 1991 to February 2011. In 10 (3.4%) of these transplants, the new methodology was applied in the preservation of the donated organ, called "Non Working Beating Heart", which is to reperfuse the coronary arteries of the graft, restoring and maintaining the beat at normothermia without its pre-and afterload during implantation.
The process of capturing and removal of the donor organ did not change, and after visual examination and palpation of possible modificatoin (atherosclerotic plaques in coronary arteries, palpable thrill, anatomical changes, etc.) cardiectomia is performed after the application of 1000 ml of 4ºC crystalloid cardioplegic solution (Celsior ®) in the aortic root, obtaining complete myocardial relaxation. The body is then placed in sterile conditions at 4 ºC for air or ground transportation.
Meanwhile, the compatible receiver is placed in the operating room, monitored and then, the native heart is exposed under general anesthesia with mechanical ventilation using transsternal median thoracotomy. At the time the donor organ arrives in the operating room, extracorporal circulation is installed into the receiver (EC) by aortic and both venae cavae cannulation. After initiation of EC and stabilization through anoxic arrest, the organ is removed, remaining stumps of right and left pulmonary veins, inferior and superior vena cava, pulmonary artery and aorta [9], maintaining the esophageal temperature around 35 ºC. Immediately before anastomosis were started, a new application of 500 ml 4ºC Celsior ® solution was made in the ascending aortic graft.
The first suture to be made is the medial half of the left pulmonary vein, followed by aortic anastomosis and, after its completion, the aortic clamping is stopped, reperfusing the coronary arteries of the graft, which spontaneously or after electrical defibrillation, resumes the beats in sinus rhythm, narrow QRS and positive T-wave. Under these conditions, the anastomosis of the pulmonary veins, inferior and superior venae cavae, and, finally, pulmonary artery are concluded, assuming, by the application of a low dose of inotropic drugs, the debt and systemic/pulmonary flow of the receiver.
With stable hemodynamic and metabolic parameters, the EC is interrupted and, after review of hemostasis and mediastinal drainage, the chest is closed in layers and the patient is referred to ICU, using breathing apparatus. In addition to routine monitoring, it is common in our service the installation of the pulmonary artery catheter for continuous control of systolic pressure (PASP), to diagnose and treat right ventricular failure possibly determined by several factors in the postoperative period.
This methodology was applied to 10 receivers, 8 male, aged 16 to 69 years (mean 40 ± 18 years) weighing 53 to 81 kg (mean 62 ± 10 kg). Regarding etiology, 4 receivers had ischemic cardiomyopathy, 2 of them dilated cardiomyopathy, 2 were presented with cardiomyopathy and 2 were valvular and chagasic.
RESULTS
The hemodynamic data in the pre-transplant evalutaion were:
• ASPo 90 to 100 (mean 96.4 ± 4.4 mmHg);
• PASP 25 to 65 (mean 50.5 ± 22.8 mmHg);
• RVP from 0.9 to 5.0 (mean 3.0 ± 1.3 Wood U);
• GTP 4.3 to 13.0 (mean 6.9 ± 2.7 mmHg).
This methodology was applied to 10 receivers, 8 male, aged 16 to 48 years (mean 28 ± 10 years) weighing 53 to 114 kg (mean 85 ± 10 kg). The causes of encephalic coma were 5 cranioencephalic traumas (CET), 4 hemorrhagic cerebrovascular accidentstrokes (HCVA) and 1 case of brain tumor.
There were no immediate deaths and all patients, both in the operating room and ICU, showed good hemodynamic, metabolic, and electrocardiographic conditions, therefore, the use of inotropic drugs was limited. The ischemia time ranged from 58 to 100 min (mean 68 ± 14 min) and EC was 90 to 140 min (mean 116 ± 19 min). The length of stay in ICU in nine patients ranged from 2 to 5 days (mean 3.4 ± 1.3 days), except one case with neurological damage, which lasted 30 days. The length of stay in the ward ranged from 7 to 36 days (mean 16 ± 10 days), and in two cases (neurological and mediastinitis), were 30 to 36 days. All patients were discharged in good clinical condition.
The late evolution ranged from 20 days to 10 months, with one death occurring 4 months after the transplant due to sepsis.
DISCUSSION
The graft preservation with the purpose of transplantation is a fundamental process due to scientific and technological advances, allowing full evaluation of the organ and its availability and various alternatives for their preservation during ischemia after removal of the donor and implantation in the receiver.
It is clear that invariably tissue damage will occur to a greater or lesser degree in the process of searching, preservation and implantation of the organ [10,11], such that the pathophysiological effects of brain death, hypoxia at the time of removal and transportation, as well as lesions due to ischemia / reperfusion are deleterious factors in the process of heart transplantation.
Taking into account the current preservation techniques, it is considered as a safe ischemia time a period between 4 to 6 hours [9.12] and longer periods are related to primary graft failure, especially if there is association with an inadequate preservation procedure.
The organic viability maintenance during graft preservation is an important prerequisite for good results and, considering the current practice of accepting organs from older, and possibly adjacent donors, the preservation techniques have become more relevant. Many teams use the hypothermic arrest, however, this method was developed when there was ample supply of younger donors and quality of the graft, and currently, with the need to extend the selection criteria, limitations to this methodology can be questioned. Maathuis et al. [13], in an elegant article, draw attention to this scenario with suggestions for new techniques of myocardial protection.
Heart surgery on a beating heart has gained ground in recent times, however, there are few studies to verify its effectiveness. Mo et al. [14] reported good results, expanding this concept little used in the past.
Osaki et al. [15] draw attention to the use of continuous myocardial perfusion for resuscitation of donor hearts, initially with blood cardioplegia at 20 ºC, and after, oxygenated blood at a temperature ranging from 20 º to 37 ºC, which somehow is applied to the method described in this study. On the other hand, keeping the heart beating during the graft preservation showed better results when compared to preservation with hypothermia and cardioplegic solution at the University of Wisconsin, extending the preservation time safely and giving surgeons better means of assessing the applicability of this organ and facilitating long distance transport [8].
Within this line of reasoning and assumption of maintaining continuous perfusion of donor heart to keep their beats, a portable apparatus was developed to preserve it and to be used during transport [7], which are monitored contractility, metabolism and vasomotor functions, extending the time preservation and avoiding injury caused by time-dependent ischemia. The potential benefits are many [16,17], since the continuous supply of oxygen and energy substrates facilitate aerobic metabolism, making the application of important hypothermia not viable. Continuous washing of toxic metabolisms occurs, which increases the graft time preservation safely, facilitating logistics, that often seems to be difficult and complex [18,19].
In 1986, we had the opportunity to publish experimental methodology for thoracic organs preservation at normothermia and in physiological conditions called heart-lung preparation [20], which included the removal all cardiopulmonary block from the donor, and was kept for up to 11 hours and then transplanted to another animal experimentation, presenting good results. This embodies the concept of keeping the beat at normothermia, and the organ preservation seems to be more consistent than the application of cardioplegic solutions and hypothermia. Brockmann et al. [21] claimed that the organ preservation by normothermic perfusion maintains the physiological pressure-flow parameters, providing more time and success in transplantation, with the possibility of increasing the number of organs donated for transplant.
In experimental research designs, there are several publications [22-26], in which heterotopic heart implant was applied taken from experimental animals and implanted in the abdomen or neck after the suture occlusion of the functions of superior and inferior venae cavae with right atrium and right and left pulmonary veins with the left atrium, in which the aortic graft is end-to-side anastomosed or with the common carotid artery, and the pulmonary artery with the abdominal portion of the inferior vena cava or the jugular, establishing a situation where the beats of the graft are maintained due to coronary perfusion and coronary sinus return directed to the venous system (Figure 1).
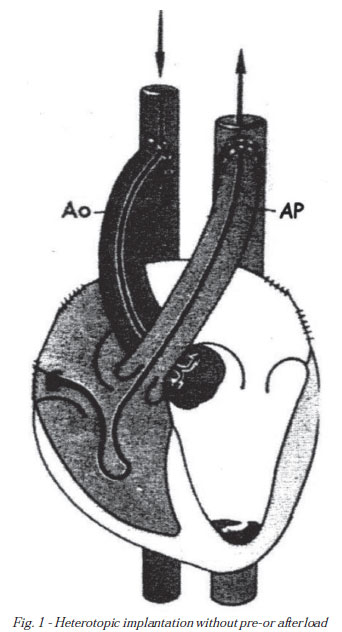
The beats are kept under these conditions, however, without pre or after-load, a process called "non working beating heart", which one of the methods of myocardial protection that is closest related to the normal physiology.
The methodology presented in this work is actually a hybrid process of preservation using hypothermic cardioplegic solution during the removal of the donor organ and transport, and also coronary reperfusion at normothermia with a beating heart without pre-or after-load while the graft implant is being performed into the receiver. The aortic anastomosis is the first one to be made and then the coronary arteries are immediately reperfused at normothermia, recovering its beating early and, these conditions are maintained throughout this procedure, allowing the graft to the possibility of an earlier recovery of contractile function, toxic metabolic elimination and positive interaction with the host, without the need for maintaining the flow and cardiac output, since the EC performs this function.
The results that draw attention for their performance was noted in this small sample (10 cases) by taking full cardiac function with good contractility and demonstrating very satisfactory postoperative parameters, with no evidence of death, low output or other clinical events.
REFERENCES
1. Pichon H, Chocron S, Alwan K, Toubin G, Kaili D, Falcoz P, et al. Crystalloid versus cold blood cardioplegia and cardiac troponin I release. Circulation. 1997;96(1):316-20. [MedLine]
2. Chocron S, Alwan K, Yan Y, Toubin G, Kaili D, Anguenot T, et al. Warm reperfusion and myocardial protection. Ann Thorac Surg. 1998;66(6):2003-7. [MedLine]
3. Lichtenstein SV, Ashe KA, el Dalati H, Cusimano RJ, Panos A, Slutsky AS. Warm heart surgery. J Thorac Cardiovasc Surg. 1991;101(2):269-74. [MedLine]
4. Wei H, Mingwu C, Wensheng W. Beating heart mitral valve replacement in 2 cases under mild hypotermia cardiopulmonary bypass. Guangxi Med Coll J. 1993;10:320-1.
5. Hui L, Wei H, Shifeng W. Aortic valve replacement on the beating heart with continuous retrograde coronary sinus perfusion. Guangxi Med Univ. 1996;13:4-6.
6. Ferrera R, Larase A, Marcsek P, Guidollet J, Verdys M, Dittmar A, et al. Comparison of different techniques of hypotermic pig heart preservation. Ann Thorac Surg. 1994;57(5):1233-9. [MedLine]
7. Hassanein WH, Zellos L, Tyrrel TA, Healey NA, Crittenden MD, Birjiniuk V, et al. Continuous perfusion of donor hearts in the beating state extends preservation time and improves recovery of function. J Thorac Cardiovasc Surg. 1998;116(5):821-30. [MedLine]
8. Lin H, Mo A, Zhang F, Huang A, Wen Z, Ling S, et al. Donor heart preservation in an empty beating state under mild hypothermia. Ann Thorac Surg. 2010;89(5):1518-23. [MedLine]
9. Dinkhuysen JJ. Transplante cardíaco ortotópico bicaval/bipulmonar. Rev Bras Cir Cardiovasc. 2003;18(3):268-72. View article
10. Belzer FO, Southard JH. Principles of solid-organ preservation by cold storage. Transplantation. 1988;45(4):673-6. [MedLine]
11. Fragomeni LS, Bonser RS, Stempfle U, Ring SW, Kaye MP, Jamieson SW. Relationship between ischemic time and post-operative performance in cardiac transplantation. Rev Bras Cir Cardiovasc. 1989;4(2):139-42. View article
12. Conte JV. Heart preservation. In: Franco KL, Verrier ED, eds. Advanced therapy in cardiac surgery. 2nd ed. Hamilton:BC Decher;2003. p.560-9.
13. Maathuis MH, Leuvenink HG, Ploeg RJ. Perspectives in organ preservation. Transplantation. 2007;83(10):1289-98. [MedLine]
14. Mo A, Lin H, Wen Z, Lu W, Long X, Zhou Y. Efficacy and safety of on-pump beating heart surgery. Ann Thorac Surg. 2008;86(6):1914-8. [MedLine]
15. Osaki S, Ishino K, Kotani Y, Honjo O, Svezawa T, Kanki K, et al. Resuscitation of non-beating donor hearts using continuous myocardical perfusion: the importance of controlled initial reperfusion. Ann Thorac Surg. 2006;81(6):2167-71. [MedLine]
16. Jacobs S, Rega F, Meyns B. Current preservation technology and future prospects of thoracic organs. Part 2: heart. Curr Opin Organ Transplant. 2010;15(2):156-9. [MedLine]
17. Cobert ML, West LM, Jessen ME. Machine perfusion for cardiac allograft preservation. Curr Opin Organ Transplant. 2008;13(5):526-30. [MedLine]
18. Ho EK, Vlad G, Colovai AI, Vasilescu ER, Schwartz J, Sondermeijer H, et al. Alloantibodies in heart transplantation. Hum Immunol. 2009;70(10):825-9. [MedLine]
19. Tenderich G, Zittermann A, Prohaska W, Koerfer R. No evidence for an improvement of long-term survival by HLA matching in heart transplant recipients. Transplant Proc. 2007;39(5):1575-9. [MedLine]
20. Dinkhuysen JJ, Souza LCB, Chaccur P, Neger F, Paes Neto F, Arnoni AS, et al. Preparado cardiopulmonar. Rev Bras Cir Cardiovasc. 1986;1(1):20-31. View article
21. Brockmann J, Reddy S, Coussios C, Pigott D, Guirriero D, Hughes D, et al. Normothermic perfusion: a new paradigm for organ preservation. Ann Surg. 2009;250(1):1-6. [MedLine]
22. Alonso DR, Starek PK, Minick CR. Studies on the pathogenesis of atheroarteriosclerosis induces in rabbit cardiac allografts by the synergy of graft rejection and hypercholesterolemia. Am J Pathol. 1977;87(2):415-42. [MedLine]
23. Foegh ML, Khirabadi BS, Nakanishi T, Vargas R, Ramwell PW. Estradiol protects against experimental cardiac transplant atherosclerosis. Transplant Proc. 1987;19(4 Suppl 5):90-5. [MedLine]
24. Clausell N, Molossi S, Sett S, Rabinovitch M. In vivo blockade of tumor necrosis factor-alpha in cholesterol-fed rabbits after cardiac transplant inhibits acute coronary artery neointimal formation. Circulation. 1994;89(6):2768-79. [MedLine]
25. Mitchell SV, Mottram PL, Purcel LJ, Dumble LJ, Millar RJ, Clunie GJ. A rabbit model for heterotopic cardiac transplantation. Transplantation. 1990;49(4):835-7. [MedLine]
26. Contreras CAM. Efeito da nanoemulsão lipídica associada a paclitaxel na prevenção da doença vascular do coração transplantado. Estudo experimental em coelhos [Tese de Doutorado]. São Paulo:Faculdade de Medicina da Universidade de São Paulo;2010. 143p.
Article receive on Monday, May 23, 2011
 All scientific articles published at rbccv.org.br are licensed under a Creative Commons license
All scientific articles published at rbccv.org.br are licensed under a Creative Commons license