ABSTRACT
We describe the successful implantation of a cardioverter-defibrillator (ICD) in two pregnant women with hypertrophic cardiomyopathy at high risk. The indication of ICD and the necessary care for ICD implantation during pregnancy are discussed and were the main objectives of this case report.
RESUMO
Descrevemos os casos de duas gestantes portadoras de cardiomiopatia hipertrófica com alto risco de morte súbita arrítmica, que foram submetidas a implante de cardioversor-desfibrilador automático (CDI) no intercurso da gestação. O momento para a realização do procedimento e os cuidados necessários para o implante do CDI durante a gestação são discutidos e foram os principais objetivos deste relato.
INTRODUCTION
Hypertrophic cardiomyopathy (HCM) is an autosomal dominant genetic trait and is characterized by left ventricular hypertrophy, especially of the interventricular septum in the absence of other conditions that justify this anatomic change [1,2].
The sudden cardiac death (SCD), with an annual incidence of 1%, is the most serious complication of this disease [2,3]. The risk of fatal arrhythmias increases, however, when: the thickness of the ventricular septum is greater than 30 mm; ventricular tachycardia is detected, the patient reports syncope or when there are sudden deaths in young relatives. The prevention of SCD with the use of automatic cardioverter defibrillator (ICD) has been recommended in high risk patients [1-3].
There are few reports of ICD implantation in pregnant women and there is no standard procedure for this condition [4]. The application of shock therapy in pregnant women with IDC, reported in two studies, had no effect on fetal development [4,5].
The aim of this report is to describe the case of two patients with HCM who underwent ICD implantation for prevention of SCD in pregnancy intercourse.
CASE REPORTS
Case 1
JMG, 24 years old, at 29 weeks' gestation, presented syncope since nine months without prodrome or associated factors. A transthoracic echocardiogram reveale HCM with ventricular septum of 29 mm and 24-hour ECG detected non-sustained ventricular tachycardia (NSVT). The patient reported she had a brother with ICD due to recovered cardiorespiratory arrest and HCM.
Case 2
TIGC, 17 years old, at 26 weeks' gestation, she had repeated episodes of syncope without prodrome or triggering factors, associated with progressive dyspnea. Transthoracic echocardiography diagnosed HCM with interventricular septum with 30 mm thick. She complained of sudden death without etiologic diagnosis in young brother and mother recently diagnosed with HCM.
ICD implantation technique
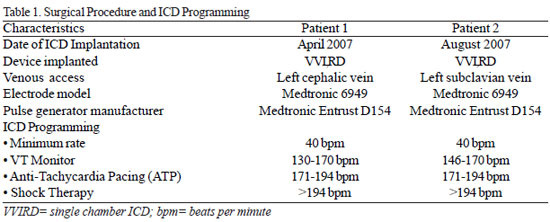
The implant procedure was performed in both cases, under intravenous sedation and local anesthesia. The abdomen of the patients was protected by blanket of lead. The transvenous lead was implanted with the aid of fluoroscopy, in the apical septum of the right ventricle and the pulse generator, housed in the left infraclavicular subcutaneous position (Figure 1). Defibrillation test was not performed.
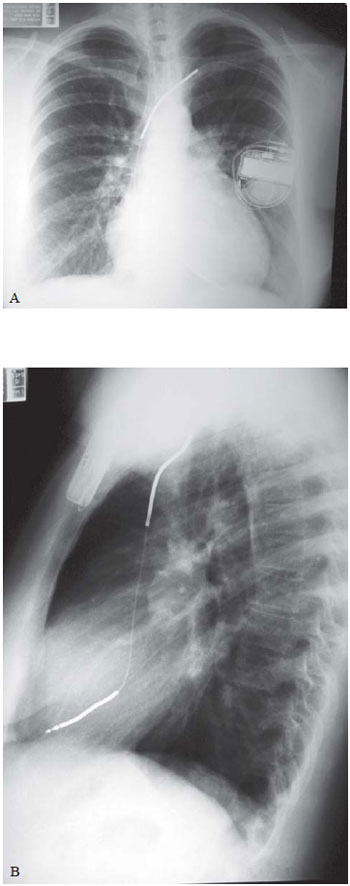
Fig. 1 - A: Chest radiograph in the anteroposterior position (A) and lateral (B), showing the position of the electrode leads of the ICD implanted through the left cephalic vein. B: Chest radiograph in the anteroposterior position (A) and lateral (B), showing the position of the electrode leads of the ICD implanted through left cephalic vein
Both pregnancies were no abnormalities. During delivery, the heart rate was monitored and shock therapies were turned off, turning the previous program of ICD after the procedure.
At the end of puerperium, patients were tested with defibrillation. Under intravenous sedation, ventricular fibrillation was induced, and in both cases, the 20J automatic shock was effective to defibrillate the heart.
During follow-up of 2.7 ± 0.2 years, there were no reports of suggestive episodes of low cerebral blood flow or heart failure. The counters of diagnoses of the ICD did not record appropriate therapy for tachycardia or ventricular fibrillation. One patient presented, however, inappropriate shock therapies for murmur detection and required replacement of the lead, which showed increased pacing impedance. There were no deficiencies or growth disorders in children of these pregnancies.
DISCUSSION
Despite its low prevalence, HCM is an important cause of SCD in young individuals [1-6]. The main risk factors for fatal arrhythmias in these patients are a history of sudden death in young relatives, the presence of syncope of unknown origin, hypotension during exercise, ventricular septal thickness of e" 30 mm and episodes of NSVT on Holter of 24 hours [1-3,6].
In this report, the diagnosis of HCM was performed during pregnancy. The identification of multiple risk factors for MSC justified the indication of using ICD during pregnancy.
In addition to the usual anesthetic care for pregnant women, two points deserve special attention: the risk of malformations by use of fluoroscopy used to guide the placement of electrode leads and the lack of knowledge of the consequences of induction of ventricular fibrillation and shock application, that are necessary to test the integrity and efficiency of the system deployed.
Among the alternatives described to prevent fetal exposure to radiation, it has been proposed the implant guided by echocardiography [7] or by electroanatomic mapping [8]. In this report, given the low risk of fetal damage from radiation in the third trimester of pregnancy, we chose to fluoroscopy, associated with the use of lead blanket on the abdomen of the pregnant women, as additional protection.
There is no evidence in the literature that the induction of ventricular fibrillation and the shocks applied to the defibrillator test during implantation cause fetal abnormalities.
The automatic application of shock in pregnant women already bearer of ICD did not cause maternal or fetal effects [4,5]. Moreover, the real need for intraoperative defibrillation test is object of controversy. Clinical studies show that in only 4% of patients shocks are ineffective in reversing ventricular fibrillation induced during ICD implantation [9,10]. These studies also showed that ineffective shocks are infrequent in patients with preserved ventricular contractility [9,10]. In the cases reported herein, we opted for no defibrillation testing.
The low risk of malformations by radiation after the first trimester of pregnancy, which allowed the use of fluoroscopy, and good ventricular function of patients which avoided the test of defibrillation, became the ICD implantation in these patients a routine procedure, despite the current pregnancy.
REFERENCES
1. Maron BJ, McKenna WJ, Danielson GK, Kappenberger LJ, Kuhn HJ, Seidman CE, et al. American College of Cardiology/European Society of Cardiology clinical expert consensus document on hypertrophic cardiomyopathy. A report of the American College of Cardiology Foundation Task Force on Clinical Expert Consensus Documents and the European Society of Cardiology Committee for Practice Guidelines. J Am Coll Cardiol. 2003;42(9):1687-713. [MedLine]
2. Maron BJ, Spirito P, Shen WK, Haas TS, Formisano F, Link MS, et al; Task Force on Clinical Expert Consensus Documents. American College of Cardiology; Committee for Practice Guidelines. European Society of Cardiology. Implantable cardioverter-defibrillators and prevention of sudden cardiac death in hypertrophic cardiomyopathy. JAMA. 2007;298(4):405-12. [MedLine]
3. Martinelli Filho M, Zimerman LI, Lorga AM, Vasconcelos JTM, Rassi A Jr, et al. Diretrizes brasileiras de dispositivos cardíacos eletrônicos implantáveis (DCEI). Arq Bras Cardiol. 2007;89(6):e210-37.
4. Natale A, Davidson T, Geiger MJ, Newby K. Implantable cardioverter-defibrillators and pregnancy: a safe combination? Circulation. 1997;96(9):2808-12. [MedLine]
5. Bonini W, Botto GL, Broffoni T, Dondina C. Pregnancy with an ICD and a documented ICD discharge. Europace. 2000;2(1):87-90. [MedLine]
6. Spirito P, Autore C, Rapezzi C, Bernabò P, Badagliacca R, Maron MS, et al. Syncope and risk of sudden death in hypertrophic cardiomyopathy. Circulation. 2009;119(13):1703-10. [MedLine]
7. Abello M, Peinado R, Merino JL, Gnoatto M, Mateos M, Silvestre J, et al. Cardioverter defibrillator implantation in a pregnant woman guided with transesophageal echocardiography. Pacing Clin Electrophysiol. 2003;26(9):1913-4. [MedLine]
8. Merino JL, Peinado R, Silvestre J. Dual-chamber implantable cardioverter defibrillator implantation guided by non-fluoroscopic electro-anatomical navigation. Europace. 2008;10(9):1124-5. [MedLine]
9. Day JD, Doshi RN, Belott P, Birgersdotter-Green U, Behboodikhah M, Ott P, et al. Inductionless or limited shock testing is possible in most patients with implantable cardioverter- defibrillators/cardiac resynchronization therapy defibrillators: results of the multicenter ASSURE Study (Arrhythmia Single Shock Defibrillation Threshold Testing Versus Upper Limit of Vulnerability: Risk Reduction Evaluation With Implantable Cardioverter-Defibrillator Implantations). Circulation. 2007;115(18):2382-9. [MedLine]
10. Lima CEB, Martinelli Filho M, Silva RT, Tamaki WT, Oliveira JC, Martins DC, et al. Portadores de CDI com limiar de desfibrilação elevado: evolução clínica e alternativas terapêuticas. Arq Bras Cardiol. 2008;90(3):177-84.
Article receive on Thursday, June 10, 2010


 All scientific articles published at rbccv.org.br are licensed under a Creative Commons license
All scientific articles published at rbccv.org.br are licensed under a Creative Commons license